Abstract
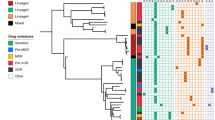
The emergence of drug-resistant tuberculosis is a major global public health threat. Thailand is one of the top 14 countries with high tuberculosis and multi-drug resistant tuberculosis rates. Immediate detection of drug-resistant tuberculosis is necessary to reduce mortality and morbidity by effectively providing treatment to ameliorate the formation of resistant strains. Limited data exist of mutation profiles in Northeastern Thailand. Here, 65 drug-resistant Mycobacterium tuberculosis isolates were used to detect mutations by polymerase chain reaction (PCR) and DNA sequencing. In the katG gene, mutations were occurred in 47 (79.7%) among 59 isoniazid resistant samples. For rpoB gene, 31 (96.9%) were observed as mutations in 32 rifampicin resistant isolates. Of 47 katG mutation samples, 45 (95.7%) had mutations in katG315 codon and 2 (4.3%) showed novel mutations at katG365 with amino acid substitution of CCG-CGG (Pro-Arg). Moreover, out of 31 rpoB mutation isolates, the codon positions rpoB516, rpoB526, rpoB531 and rpoB533 were 3 (9.7%), 8 (25.8%), 11 (35.5%) and 1 (3.2%), respectively. Seven isolates of double point mutation were found [rpoB516, 526; 1 (3.2%) and rpoB516, 531; 6 (19.4%)]. In addition, 1 (3.2%) sample had triple point mutation at codon positions rpoB516, 526 and 531. Common and novel mutation codons of the rpoB and katG genes were generated. Although DNA sequencing showed high accuracy, conventional PCR could be applied as an initial marker for screening drug-resistant Mycobacterium tuberculosis isolates in limit resources region. Mutations reported here should be considered when developing new molecular diagnostic methods for implementation in Northeastern Thailand.


Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Ahmed M, Ahmed AR, Mohammed S (2013) First experience with using simple polymerase chain reaction-based methods as an alternative to phenotypic drug susceptibility testing for Myobacterium tuberculosis in Iraq. Int J Appl Basic Med Res 3:98–105. https://doi.org/10.4103/2229-516x.117069
Allegui Z, Ghariani A, Draoui H et al (2012) Detection of isoniazid and rifampin resistance of Mycobacterium tuberculosis by a multiplex allele-specific polymerase chain reaction (PCR) assay. Int J Mycobacteriol 1:34–39. https://doi.org/10.1016/j.ijmyco.2012.01.006
Ando H, Kondo Y, Suetake T et al (2010) Identification of katG mutations associated with high-level isoniazid resistance in Mycobacterium tuberculosis. Antimicrob Agents Chemother 54:1793–1799. https://doi.org/10.1128/AAC.01691-09
Anukool U, Phunpae P, Tharinjaroen CS et al (2020) Genotypic distribution and a potential diagnostic assay of multidrug-resistant tuberculosis in northern thailand. Infect Drug Resist 13:3375–3382. https://doi.org/10.2147/IDR.S263082
Aparna Lingala ML, Srikantam A, Jain S et al (2010) Clinical and geographical profiles of rpoB gene mutations in Mycobacterium tuberculosis isolates from Hyderabad and Koraput in India. J Microbiol Antimicrob 2:13–18
Boonaiam S, Chaiprasert A, Prammananan T, Leechawengwongs M (2010) Genotypic analysis of genes associated with isoniazid and ethionamide resistance in MDR-TB isolates from Thailand. Clin Microbiol Infect 16:396–399. https://doi.org/10.1111/j.1469-0691.2009.02838
Caws M, Minh Duy P, Quang Tho D et al (2006) Mutations prevalent among Rifampin- and isoniazid-resistant Mycobacterium tuberculosis isolates from a hospital in Vietnam. J Clin Microbiol 44:2333–2337. https://doi.org/10.1128/JCM.00330-06
Chaves F, Alonso-Sanz M, Rebollo MJ et al (2000) rpoB mutations as an epidemiologic marker in rifampin-resistant Mycobacterium tuberculosis. Int J Tuberc Lung Dis 4(8):765–770
Cheng S, Hide M, Pheng SH et al (2021) Resistance to second-line anti-TB drugs in Cambodia: a phenotypic and genetic study. Infect Drug Resist 14:1089–1104. https://doi.org/10.2147/idr.s289907
Fan XY, Hu ZY, Xu FH et al (2003) Rapid detection of rpoB gene mutations in rifampin-resistant Mycobacterium tuberculosis isolates in Shanghai by using the amplification refractory mutation system. J Clin Microbiol 41:993–997. https://doi.org/10.1128/JCM.41.3.993-997.2003
Farnia P, Taghavi K, Sh S, et al (2010) Susceptibility testing multiplex - PCR, PCR SSCP and PCR ELISA Mycobacteriology Research Center (MRC), 2168099; Tehran: National Research Institute of Tuberculosis and Lung Diseases, 978–600. https://doi.org/10.13140/RG.2.2.14291.07200
Genestet C, Hodille E, Berland JL et al (2020) Whole-genome sequencing in drug susceptibility testing of Mycobacterium tuberculosis in routine practice in Lyon. France Int J Antimicrob Agents 55:105912. https://doi.org/10.1016/j.ijantimicag.2020.105912
González N, Torres MJ, Aznar J, Palomares JC (1999) Molecular analysis of rifampin and isoniazid resistance of Mycobacterium tuberculosis clinical isolates in Seville, Spain. Tuber Lung Dis 79:187–190. https://doi.org/10.1054/tuld.1998.0195
Gupta A, Prakash P, Singh SK, Anupurba S (2013) Rapid Genotypic Detection of rpoB and katG Gene Mutations in Mycobacterium tuberculosis Clinical Isolates from Northern India as Determined by MAS-PCR. J Clin Lab Anal 27:31–37. https://doi.org/10.1002/jcla.21558
Hang NTL, Hijikata M, Maeda S et al (2019) Whole genome sequencing, analyses of drug resistance-conferring mutations, and correlation with transmission of Mycobacterium tuberculosis carrying katG-S315T in Hanoi, Vietnam. Sci Rep 9:15354. https://doi.org/10.1038/s41598-019-51812-7
Isakova J, Sovkhozova N, Vinnikov D et al (2018) Mutations of rpoB, katG, inhA and ahp genes in rifampicin and isoniazid-resistant Mycobacterium tuberculosis in Kyrgyz Republic. BMC Microbiol 18:1–8. https://doi.org/10.1186/s12866-018-1168-x
Ismail N-A, Fazli Ismail M, Suraiya S et al (2016) Genotypic detection of rpoB and katG gene mutations associated with rifampicin and isoniazid resistance in Mycobacterium tuberculosis isolates: a local scenario (Kelantan). Malays J Med Sci 23(1):22–26
Jaiswal I, Jain A, Singh P et al (2017) Mutations in katG and inhA genes of isoniazid-resistant and -sensitive clinical isolates of Mycobacterium tuberculosis from cases of pulmonary tuberculosis and their association with minimum inhibitory concentration of isoniazid. Clin Epidemiol Glob Heal 5:143–147. https://doi.org/10.1016/j.cegh.2016.08.008
Jaksuwan R, Tharavichikul P, Patumanond J et al (2017) Genotypic distribution of multidrug-resistant and extensively drug-resistant tuberculosis in northern Thailand. Infect Drug Resist 10:167–174. https://doi.org/10.2147/IDR.S130203
Jang JG, Chung JH (2020) Diagnosis and treatment of multidrug-resistant tuberculosis. Yeungnam Univ J Med 37:277–285. https://doi.org/10.12701/yujm.2020.00626
Jena L, Waghmare P, Kashikar S et al (2014) Computational approach to understanding the mechanism of action of isoniazid, an anti-TB drug. Int J Mycobacteriol 3:276–282. https://doi.org/10.1016/j.ijmyco.2014.08.003
Jintaridh P, Ramasoota P, Pyar K et al (2014) Novel mutation detection in rpoB of Rifampicin-resistant Mycobacterium tuberculosis using pyrosequencing. Southeast Asian J Trop Med Public Health 45:843–852
Jureen P, Engstrand L, Eriksson S et al (2006) Rapid detection of rifampin resistance in Mycobacterium tuberculosis by pyrosequencing technology. J Clin Microbiol 44:1925–1929. https://doi.org/10.1128/JCM.02210-05
Lange C, Aarnoutse RE, Alffenaar JWC et al (2019) Management of patients with multidrug-resistant tuberculosis. Int J Tuberc Lung Dis 23:645–662. https://doi.org/10.5588/ijtld.18.0622
Lee ASG, Lim IHK, Tang LLH et al (1999) Contribution of kasA analysis to detection of isoniazid-resistant Mycobacterium tuberculosis in Singapore. Antimicrob Agents Chemother 43:2087–2089. https://doi.org/10.1128/aac.43.8.2087
Lee ASG, Lim IHK, Tang LLH, Sin YW (2005) High frequency of mutations in the rpoB gene in rifampin-resistant clinical isolates of Mycobacterium tuberculosis from Singapore. J Clin Microbiol 43:2026–2027
Li MC, Chen R, Lin SQ et al (2020) Detecting ethambutol resistance in Mycobacterium tuberculosis isolates in China: a comparison between phenotypic drug susceptibility testing methods and DNA sequencing of embAB. Front Microbiol 11:1–7. https://doi.org/10.3389/fmicb.2020.00781
Liu L, Jiang F, Chen L et al (2018) The impact of combined gene mutations in inhA and ahpC genes on high levels of isoniazid resistance amongst katG non-315 in multidrug-resistant tuberculosis isolates from China. Emerg Microbes Infect 7:183. https://doi.org/10.1038/s41426-018-0184-0
Makadia JS, Jain A, Patra SK et al (2012) Emerging trend of mutation profile of rpoB gene in MDR tuberculosis, North India. Indian J Clin Biochem 27:370–374. https://doi.org/10.1007/s12291-012-0228-5
Mani C, Selvakumar N, Narayanan S, Narayanan PR (2001) Mutations in the rpoB gene of multidrug-resistant Mycobacterium tuberculosis clinical isolates from India. J Clin Microbiol 39:2987–2990. https://doi.org/10.1128/JCM.39.8.2987-2990.2001
Mistri SK, Sultana M, Kamal SMM et al (2016) Evaluation of efficiency of nested multiplex allele-specific PCR assay for detection of multidrug resistant tuberculosis directly from sputum samples. Lett Appl Microbiol 62:411–418. https://doi.org/10.1111/lam.12564
Nimri L, Samara H, Batchoun R (2011) Detection of mutations associated with multidrug-resistant Mycobacterium tuberculosis clinical isolates. FEMS Immunol Med Microbiol 62:321–327. https://doi.org/10.1111/j.1574-695X.2011.00814.x
Nurpermatasari A, Harahap U, Siagian P (2018) Identification gene mutations rpoB cause of multidrug resistance tuberculosis in Haji Adam Malik hospital. Asian J Pharm Clin Res 11:155–158. https://doi.org/10.22159/ajpcr.2018.v11s1.26595
Palomino JC, Martin A (2014) Drug resistance mechanisms in Mycobacterium tuberculosis. Antibiotics 3:317–340
Pandey S, Lamichhane A, Byanjankar A et al (2017) Direct detection of rpoB and katG gene mutations of Mycobacterium tuberculosis in clinical samples. Asian Pac J Trop Biomed 7:698–701. https://doi.org/10.1016/j.apjtb.2017.07.018
Pang Y, Lu J, Wang Y et al (2013) Study of the rifampin monoresistance mechanism in Mycobacterium tuberculosis. Antimicrob Agents Chemother 57:893–900. https://doi.org/10.1128/AAC.01024-12
Prammananan T, Cheunoy W, Taechamahapun D et al (2008) Distribution of rpoB mutations among multidrug-resistant Mycobacterium tuberculosis (MDRTB) strains from Thailand and development of a rapid method for mutation detection. Clin Microbiol Infect 14:446–453. https://doi.org/10.1111/j.1469-0691.2008.01951
Qian L, Abe C, Lin TP et al (2002) rpoB genotypes of Mycobacterium tuberculosis Beijing family isolates from East Asian countries. J Clin Microbiol 40:1091–1094. https://doi.org/10.1128/JCM.40.3.1091-1094.2002
Ravibalan T, Samrot AV, Maruthai K et al (2015) Evaluation of multiplex polymerase chain reaction assay for the detection of katG (S315T) gene mutation in Mycobacterium tuberculosis isolates from Puducherry, South India. J Pure Appl Microbiol 9:2339–2345
Rueangsak K, Tanuchit S, Chumpol J et al (2020) 24-locus MIRU-VNTR and Spoligotyping analysis of drug-resistant Mycobacterium tuberculosis strains isolated from Northeastern Thailand. Infect Genet Evol 85:104449. https://doi.org/10.1016/j.meegid.2020.104449
Seifert M, Catanzaro D, Catanzaro A, Rodwell TC (2015) Genetic mutations associated with isoniazid resistance in Mycobacterium tuberculosis: a systematic review. PLoS ONE 10:1–13. https://doi.org/10.1371/journal.pone.0119628
Suthum K, Samosornsuk W, Samosornsuk S (2020) Characterization of katG, inhA, rpoB and pncA in Mycobacterium tuberculosis isolates from MDR-TB risk patients in Thailand. J Infect Dev Ctries 14:268–276. https://doi.org/10.3855/jidc.11974
Tajbakhsh A, Ghasemi F, Mirbagheri SZ et al (2018) Investigation of the rpoB mutations causing Rifampin resistance by rapid screening in Mycobacterium tuberculosis in North-East of Iran. Iran J Pathol 13(4):429–437
Timmins GS, Deretic V (2006) Mechanisms of action of isoniazid. Mol Microbiol 62:1220–1227
Traore H, Fissette K, Bastian I et al (2000) Detection of rifampicin resistance in Mycobacterium tuberculosis isolates from diverse countries by a commercial line probe assay as an initial indicator of multidrug resistance. Int J Tuberc Lung Dis 4(5):481–484
Ullah I, Ahmad W, Shah AA et al (2019) Detection of rifampicin resistance of Mycobacterium tuberculosis using multiplex allele specific polymerase chain reaction (MAS-PCR) in Pakistan. Infect Genet Evol 71:42–46. https://doi.org/10.1016/j.meegid.2019.03.007
Valvatne H, Syre H, Kross M et al (2009) Isoniazid and rifampicin resistance-associated mutations in Mycobacterium tuberculosis isolates from Yangon, Myanmar: implications for rapid molecular testing. J Antimicrob Chemother 64:694–701. https://doi.org/10.1093/jac/dkp292
Van Embden JDA, Cave MD, Crawford JT et al (1993) Strain Identification of Mycobacterium tuberculosis by DNA fingerprinting: recommendations for a standardized methodology. J Clin Microbiol 31:406–409
Van Soolingen D, De Haas PEW, Rogier Van Doorn H et al (2000) Mutations at amino acid position 315 of the katG gene are associated with high-level resistance to isoniazid, other drug resistance, and successful transmission of Mycobacterium tuberculosis in The Netherlands. J Infect Dis 182:1788–1790
Williams DL, Waguespack C, Eisenach K et al (1994) Characterization of Rifampin resistance in pathogenic mycobacteria. Antimicrob Agents Chemother 38:2380–2386
World Health Organizatin (2020) . Global tuberculosis report 2020. http://apps.who.int/bookorders. Accessed 11 Mar 2021.
Wu XQ, Lu Y, Zhang JX et al (2006) Detection of the mutations in katG315 and inhA-15 of Mycobacterium tuberculosis strains isolated from Chinese patients. Chin Med J (engl) 119:230–233. https://doi.org/10.1097/00029330-200602010-00011
Yue J, Shi W, Xie J et al (2003) Mutations in the rpoB gene of multidrug-resistant Mycobacterium tuberculosis isolates from China. J Clin Microbiol 41:2209–2212. https://doi.org/10.1128/JCM.41.5.2209-2212.2003
Zhang Y, Heym B, Allen B et al (1992) The catalase—peroxidase gene and isoniazid resistance of Mycobacterium tuberculosis. Nature 358:591–593. https://doi.org/10.1038/358591a0
Acknowledgements
This research project was supported by Khon Kaen University Research Grant (Project Numbers 590038 and 6100033) and Centre for Research and Development of Medical Diagnostic Laboratories, Khon Kaen University. We would like to thank the KKU Scholarship for ASEAN and GMS Countries’ Personnel of Academic Year 2019, Khon Kaen University for providing a scholarship for Ei Phoo Thwe. Special thanks also go to Miss Kulrattana Rueangsak for her technical assistance.
Funding
The present study was supported by Centre for Research and Development in Medical Diagnostic Laboratory, Faculty of Associated Medical Sciences, Khon Kaen University and the Graduate School, Khon Kaen University, Thailand.
Author information
Authors and Affiliations
Contributions
Not applicable.
Corresponding author
Ethics declarations
Conflict of interest
The author declares that they have no conflict of interest.
Ethical approval
This study was approved by Khon Kaen University Ethics Committee for Human Research (HE631314).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Thwe, E.P., Namwat, W., Pinlaor, P. et al. Novel mutations detected from drug resistant Mycobacterium tuberculosis isolated from North East of Thailand. World J Microbiol Biotechnol 37, 194 (2021). https://doi.org/10.1007/s11274-021-03163-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-021-03163-7