Abstract
Background
Despite controversy regarding the role of neoadjuvant chemotherapy (NAC) in pancreatic adenocarcinoma, nearly half of resected patients do not receive chemotherapy postoperatively. This study aimed to examine whether use of NAC compensates for omission of adjuvant chemotherapy (AC) for resected pancreatic adenocarcinoma.
Methods
Adults with resected stages 1 to 3 pancreatic adenocarcinoma were enrolled from the National Cancer Database NCDB (2006–2016). Overall survival (OS) analyses were used to examine the impact of NAC on those who did not receive AC.
Results
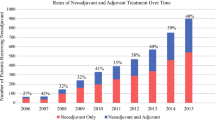
The study analyzed a national cohort of 56,286 patients: 30% without chemotherapy, 11% with NAC, 54% with AC, and 5% with NAC plus AC. Use of NAC increased by more than 400% from 2006 to 2016, whereas the rates for omission of chemotherapy remained unchanged. The OS rates were similar between the patients who received NAC and those who received AC (hazard ratio, 0.97; p = 0.21). Among the patients who did not receive AC, NAC was associated with improved OS (26.7 vs. 18.4 months; p < 0.0001). The patients who did not receive AC but underwent NAC had a median OS comparable with the OS of those who received AC alone (26.9 vs. 24.7 months). In the adjusted analysis, the use of NAC for those without AC was significantly associated with improved OS (estimate, − 0.24; p < 0.0001).
Conclusions
Although data are limited regarding the survival benefit derived from neoadjuvant versus adjuvant chemotherapy in pancreatic adenocarcinoma, nearly half of patients do not receive adjuvant chemotherapy. This study demonstrates that the use of NAC lessens the survival disadvantage caused by omission of AC. Despite controversy, NAC may be considered for pancreatic adenocarcinoma patients given the high likelihood that adjuvant chemotherapy will be omitted.


Similar content being viewed by others
References
American Cancer Society: Cancer Facts & Figures 2019. Retrieved 27 December 2019 at https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2019/cancer-facts-and-figures-2019.pdf.
National Comprehensive Cancer Network: NCCN Guidelines, version 1.2020, Pancreatic Adenocarcinoam. Retrieved 26 December 2019 at https://www.nccn.org/professionals/physician_gls/pdf/pancreatic.pdf.
Cameron JL, He J. Two thousand consecutive pancreaticoduodenectomies. J Am Coll Surg. 2015;220:530–6.
Zureikat AH, Postlewait LM, Liu Y, et al. A multi-institutional comparison of perioperative outcomes of robotic and open pancreaticoduodenectomy. Ann Surg. 2016;264:640–9.
Huang JJ, Yeo CJ, Sohn TA, et al. Quality of life and outcomes after pancreaticoduodenectomy. Ann Surg. 2000;231:890–8.
Schniewind B, Bestmann B, Henne-Bruns D, Faendrich F, Kremer B, Kuechler T. Quality of life after pancreaticoduodenectomy for ductal adenocarcinoma of the pancreatic head. Br J Surg. 2006;93:1099–107.
Kalser MH, Ellenberg SS. Pancreatic cancer: adjuvant combined radiation and chemotherapy following curative resection. Arch Surg. Chicago. IL 1960. 1985;120:899–903.
Neoptolemos JP, Stocken DD, Friess H, et al. A randomized trial of chemoradiotherapy and chemotherapy after resection of pancreatic cancer. N Engl J Med. 2004;350:1200–10.
Oettle H, Neuhaus P, Hochhaus A, et al. Adjuvant chemotherapy with gemcitabine and long-term outcomes among patients with resected pancreatic cancer: the CONKO-001 randomized trial. JAMA. 2013;310:1473–81.
Neoptolemos JP, Palmer DH, Ghaneh P, et al. Comparison of adjuvant gemcitabine and capecitabine with gemcitabine monotherapy in patients with resected pancreatic cancer (ESPAC-4): a multicentre, open-label, randomised, phase 3 trial. Lancet London England. 2017;389:1011–24.
Mackay TM, Smits FJ, Roos D, et al. The risk of not receiving adjuvant chemotherapy after resection of pancreatic ductal adenocarcinoma: a nationwide analysis. HPB. 2020;22(2):233–40.
Kagedan DJ, Dixon ME, Raju RS, et al. Predictors of adjuvant treatment for pancreatic adenocarcinoma at the population level. Curr Oncol Toronto Ontario. 2016;23:334–42.
Lim JE, Chien MW, Earle CC. Prognostic factors following curative resection for pancreatic adenocarcinoma: a population-based, linked database analysis of 396 patients. Ann Surg. 2003;237:74–85.
Russ AJ, Weber SM, Rettammel RJ, Mahvi DM, Rikkers LF, Cho CS. Impact of selection bias on the utilization of adjuvant therapy for pancreas adenocarcinoma. Ann Surg Oncol. 2010;17:371–6.
Merkow RP, Bilimoria KY, Tomlinson JS, et al. Postoperative complications reduce adjuvant chemotherapy use in resectable pancreatic cancer. Ann Surg. 2014;260:372–7.
Tzeng CW, Tran Cao HS, Lee JE, et al. Treatment sequencing for resectable pancreatic cancer: influence of early metastases and surgical complications on multimodality therapy completion and survival. J Gastrointest Surg. 2014;18:16–24.
Unno M, Motoi F, Matsuyama Y, et al. Randomized phase II/III trial of neoadjuvant chemotherapy with gemcitabine and S-1 versus upfront surgery for resectable pancreatic cancer (Prep-02/JSAP-05). J Clin Oncol. 2019;37(4 Suppl):189–189.
Hashmi A, Kozick Z, Fluck M, et al. Neoadjuvant versus adjuvant chemotherapy for resectable pancreatic adenocarcinoma: a National Cancer Database analysis. Am Surg. 2018;84:1439–45.
Lutfi W, Talamonti MS, Kantor O, et al. Perioperative chemotherapy is associated with a survival advantage in early-stage adenocarcinoma of the pancreatic head. Surgery. 2016;160:714–24.
Deyo RA, Cherkin DC, Ciol MA. Adapting a clinical comorbidity index for use with ICD-9-CM administrative databases. J Clin Epidemiol. 1992;45:613–9.
van der Geest LG, van Rijssen LB, Molenaar IQ, et al. Volume-outcome relationships in pancreatoduodenectomy for cancer. HPB. 2016;18:317–24.
Versteijne E, Suker M, Groothuis K, et al. Preoperative chemoradiotherapy versus immediate surgery for resectable and borderline resectable pancreatic cancer: results of the Dutch randomized phase 3 PREOPANC Trial. J Clin Oncol. 0(0):JCO.19.02274.
Paniccia A GA, Zenati MS, AlAbbas AI, Jung JP, Bahary N, et al. Predictors of disease progression or performance status decline in patients undergoing neoadjuvant therapy for localized pancreatic head adenocarcinoma. Ann Surg Oncol. In press.
Nussbaum DP, Adam MA, Youngwirth LM, et al. Minimally invasive pancreaticoduodenectomy does not improve use or time to initiation of adjuvant chemotherapy for patients with pancreatic adenocarcinoma. Ann Surg Oncol. 2016;23:1026–33.
Tran Cao HS, Lopez N, Chang DC, et al. Improved perioperative outcomes with minimally invasive distal pancreatectomy: results from a population-based analysis. JAMA Surg. 2014;149:237–43.
Adam MA, Choudhury K, Goffredo P, et al. Minimally invasive distal pancreatectomy for cancer: short-term oncologic outcomes in 1733 patients. World J Surg. 2015;39:2564–72.
Liu JB, Berian JR, Liu Y, Cohen ME, Ko CY, Hall BL. Procedure-specific trends in surgical outcomes. J Am Coll Surg. 2018;226:30–36.e34.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
There are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Adam, M.A., Nassour, I., Hoehn, R. et al. Neoadjuvant Chemotherapy for Pancreatic Adenocarcinoma Lessens the Deleterious Effect of Omission of Adjuvant Chemotherapy. Ann Surg Oncol 28, 3800–3807 (2021). https://doi.org/10.1245/s10434-020-09446-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-020-09446-x