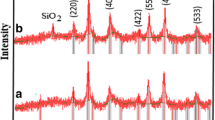
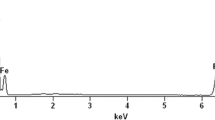
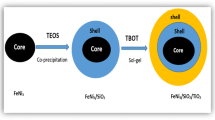
Abstract
The formation kinetics of magnetite nanoparticles upon their stabilization with potassium humate (PH) has been studied. It has been shown that the properties of Fe3O4/PH composite particles depend essentially on the time interval between the onset of nucleation and the moment of PH addition, as well as on PH concentration during the synthesis. It has been found that the introduction of PH in the optimum concentration gives rise to the formation of a protective adsorption layer, which provides magnetic particles with resistance to aggregation and sedimentation. The study of the sorption properties of magnetic Fe3O4/PH particles with respect to an antibiotic, ciprofloxacin, as depending on the synthesis conditions and characteristics of the particles has shown that the composite particles remove the antibiotic from aqueous solutions much more efficiently than do the unmodified magnetite particles.







Similar content being viewed by others
REFERENCES
Fraga García, P., Brammen, M., Wolf, M., Reinlein, S., Freiherr von Roman, M., and Berensmeier, S., Sep. Purif. Technol., 2015, vol. 150, p. 29.
Roth, H.-C., Schwaminger, S.P., Peng, F., and Berensmeier, S., Chem.Open, 2016, p. 183.
Bauer, L.M., Situ, S.F., Griswold, M.A., and Samia, A.C.S., Nanoscale, 2016, vol. 8, p. 12162.
Colombo, M., Romero, S.C., Casula, M.F., Gutierrez, L., Morales, M.P., Bohm, I.B., Heverhagen, J.T., Prosperi,PH D., and Parak, W.J., Chem. Soc. Rev., 2012, vol. 41, p. 4306.
Tombácz, E., Turcu, R., Socoliuc, V., and Vekas, L., Biochem. Biophys. Res. Commun., 2015, vol. 468, p. 442.
Rossi, L.M., Costa, N.J.S., Silva, F.P., and Wojcieszak, R., Green Chem., 2014, vol. 16, p. 2906.
Tian, L.-L., Zhang, M.-J., Wu, C., Wei, Y., Zheng, J.-X., and Lin, L.-P., ACS Appl. Mater. Interfaces, 2015, vol. 7, p. 26284.
Li, Z., Lowry, G.V., Fan, J., Liu, F., and Chen, J., Sci. Total Environ., 2018, vol. 628, p. 177.
Verlicchi, P., Al Aukidy, M., and Zambello, E., Sci. Total Environ., 2012, vol. 429, p. 123.
Massart, R., IEEE Trans. Magn., 1981, vol. 17, p. 1247.
Kydralieva, K.A., Yurishcheva, A.A., Dzhardimalieva, G.I., and Jorobekova, S.J., J. Inorg. Organomet. Polym. Mater., 2016, vol. 26, p. 1212.
Pomogailo, A.D., Rozenberg, A.S., and Uflyand, I.E., Metallopolimernye nanokompozity (Metal Polymer Nanocomposites), Moscow: Khimiya, 2000.
Irzhak, V.I., Rev. J. Chem., 2016, vol. 6, p. 370.
Descamps, M. and Willart, J.-F., Int. J. Pharm., 2018, vol. 542, p. 186.
Gubin, S.P., Koksharov, Yu.A., Khomutov, G.B., and Yurkov, G.Yu., Usp. Khim., 2003, vol. 74, p. 539.
Munoz, M., Pedro, Z.M., Casas, J.A., and Rodriguez, J.J., Appl. Catal. B, 2015, vol. 176, p. 249.
Su, C., J. Hazard. Mater., 2017, vol. 322, p. 48.
Philippe, A. and Schaumann, G.E., Environ. Sci. Technol., 2014, vol. 48, p. 8946.
Aleksashkin, I.V., Pershina, E.D., and Kazdobin, K.A., Uchen. Zapiski Tavr. Nats. Univ. im. V.I. Vernadskogo, Ser. Biol.,Khim., 2010, vol. 23, no. 3, p. 227.
http://www.micromeritics.com/Pressroom/Press-Release-List/Micromeritics-Analytical-Services-MAS-Reports-Average-Particle-Size-of-Nanoparticles.aspx.
Kumarage, W.G.C., Wijesundera, R.P., Seneviratne, V.A., Jayalath, C.P., Vargá, T., Nandasiri, M.I., and Dassanayake, B., Mater. Chem. Phys., 2017, vol. 200, p. 1.
Zhorobekova, Sh.Zh., Makroligandnye svoistva guminovykh kislot (Macroligand Properties of Humic Acids), Bishkek: Ilim, 1987.
Nuzzo, A., Sánchez, A., Fontaine, B., and Piccolo, A., J. Geochem. Explor., 2013, vol. 129, p. 1.
Illés, E. and Tombácz, E., J. Colloid Interface Sci., 2006, vol. 295, p. 115.
Kokorin, A.I., Kulyabko, L.S., Degtyarev, E.N., Kovarskii, A.L., Patsaeva, S.V., Dzhardimalieva, G.I., Yurishcheva, A.A., and Kydralieva, K.A., Russ. J. Phys. Chem. B, 2018, vol. 12, no. 1, p. 172.
Illés, E. and Tombácz, E., Colloids Surf. A, 2003, vol. 230, p. 99.
Yang, K., Lin, D.H., and Xing, B.S., Langmuir, 2009, vol. 25, p. 3571.
Hur, J. and Schlautman, M.A., J. Colloid Interface Sci., 2004, vol. 277, p. 264.
Esmaeili, H., Ebrahimi, A., Hajian, M., and Pourzamani, H.R., Int. J. Environ. Health Eng., 2012, vol. 1, p. 33.
Brunauer, S., Emmett, P.H., and Teller, E., J. Am. Chem. Soc., 1938, vol. 60, p. 309.
Lippens, B.C. and De Boer, J.H., J. Catal., 1965, vol. 4, p. 319.
Barrett, E.P., Joyner, L.G., and Halenda, P.P., J. Am. Chem. Soc., 1951, vol. 73, p. 373.
Ahmed, M.B., Zhou, J.L., Ngo, H.H., and Guo, W., Sci. Total Environ., 2015, vol. 532, p. 112.
Gu, C. and Karthikeyan, K.G., Environ. Sci. Technol., 2005, vol. 39, p. 9166.
ACKNOWLEDGMENTS
We are very grateful to Cand. Chem. A.V. Sybachin (Department of Macromolecular Compounds, Moscow State University) for help in studying colloid properties of nanoparticles.
Funding
This work was supported by the Russian Foundation for Basic Research, projects nos. 18-33-01270/18 and 17-43-500631 and performed within the framework of a state order to the Institute of Problems of Chemical Physics, Russian Academy of Sciences (0089-2019-0008). Sorption properties of materials with respect to ciprofloxacin were studied within the framework of a state order to the Institute of Chemistry, Far East Branch, Russian Academy of Sciences (265-2019-0002).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of intere-st.
Additional information
Translated by L. Tkachenko
Rights and permissions
About this article
Cite this article
Dzhardimalieva, G.I., Irzhak, V.I., Bratskaya, S.Y. et al. Stabilization of Magnetite Nanoparticles in Humic Acid Medium and Study of Their Sorption Properties. Colloid J 82, 1–7 (2020). https://doi.org/10.1134/S1061933X20010032
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061933X20010032