Abstract
Background
Body composition metrics as predictors of adverse events are a growing area of interest in oncology research. One barrier to the use of these metrics in clinical practice is the lack of standardized cut points for identifying patients with at-risk body composition profiles. We examined the association of chemotherapy adverse events with several body composition measures, using alternative cut points from published studies.
Methods
This is a retrospective study of women diagnosed with early breast cancer (EBC). Axial computerized tomography (CT) images from lumbar L3 segments were analyzed for the following body composition measures: myosteatosis (low Skeletal Muscle Density/SMD), sarcopenia (low Skeletal Muscle Index/SMI), and high Visceral Adipose Tissue (VAT). Adverse events during chemotherapy were dose reduction, early treatment discontinuation, and hospitalization. Log-binomial modeling was used to evaluate associations between body composition measures at different cut points with adverse events, adjusting for age, race, Body Mass Index/BMI, and comorbidities. Relative risks were reported as the measure of association.
Results
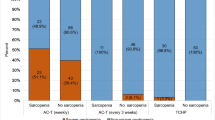
In a sample of 338 women, mean age was 51, 14% were age 65 or older, 32% were non-white, 40% had obesity (/BMI ≥ 30 kg/m2), and mean number of comorbidities was 1.56. In multivariable analysis (MV), all three SMD cut points for myosteatosis had significant associations with total number of adverse events, as well as different cut points having significant associations with either dose reduction, early treatment discontinuation or hospitalization. SMI and VAT were not significant in the MV analysis; however, in some models, age and total comorbidities were significant for adverse events.
Conclusions
Among CT-derived measures of body composition, myosteatosis determined at any of three SMD cut points was associated with total and individual adverse events during chemotherapy for early breast cancer.
Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A (2016) Cancer statistics, 2016. CA Cancer J Clin 66:7–30
Torre LA, Siegel RL, Ward EM et al (2016) Global cancer incidence and mortality rates and trends–an update. Cancer Epidemiol Biomark Prev 25:16–27
Surveilance epidemiology and end results program. Breast cancer epidemiology (2019). https://seer.cancer.gov
Veronesi U, Marubini E, Del Vecchio M et al (1995) Local recurrences and distant metastases after conservative breast cancer treatments: partly independent events. J Natl Cancer Inst 87:19–27
Gobbini E, Ezzalfani M, Dieras V et al (2018) Time trends of overall survival among metastatic breast cancer patients in the real-life ESME cohort. Eur J Cancer 96:17–24
Abdel-Rahman O (2019) Outcomes of early-stage breast cancer patients treated with sequential anthracyclines-taxanes in relationship to relative dosing intensity: a secondary analysis of a randomized controlled trial. Clin Transl Oncol 21:239–245
Horowitz NS, Wright AA (2015) Impact of obesity on chemotherapy management and outcomes in women with gynecologic malignancies. Gynecol Oncol 138:201–206
Griggs JJ, Sorbero ME, Lyman GH (2005) Undertreatment of obese women receiving breast cancer chemotherapy. Arch Intern Med 165:1267–1273
Lyman GH (2012) Weight-based chemotherapy dosing in obese patients with cancer: back to the future. J Oncol Pract 8:e62–e64
Furlanetto J, Eiermann W, Marme F et al (2016) Higher rate of severe toxicities in obese patients receiving dose-dense (dd) chemotherapy according to unadjusted body surface area: results of the prospectively randomized GAIN study. Ann Oncol 27:2053–2059
Gouerant S, Leheurteur M, Chaker M et al (2013) A higher body mass index and fat mass are factors predictive of docetaxel dose intensity. Anticancer Res 33:5655–5662
Caan BJ, Cespedes Feliciano EM, Prado CM et al (2018) Association of muscle and adiposity measured by computed tomography with survival in patients with nonmetastatic breast cancer. JAMA Oncol 4:798–804
Cespedes Feliciano EM, Chen WY, Lee V et al (2019) Body composition, adherence to anthracycline and taxane-based chemotherapy, and survival after nonmetastatic breast cancer. JAMA Oncol 6:264
Aleixo GFP, Williams GR, Nyrop KA et al (2019) Muscle composition and outcomes in patients with breast cancer: meta-analysis and systematic review. Breast Cancer Res Treat 177:569
Caan BJ, Cespedes Feliciano EM, Kroenke CH (2018) The importance of body composition in explaining the overweight paradox in cancer-counterpoint. Cancer Res 78:1906–1912
Shachar SS, Williams GR, Muss HB et al (2016) Prognostic value of sarcopenia in adults with solid tumours: a meta-analysis and systematic review. Eur J Cancer 57:58–67
Rosenberg IH (1997) Sarcopenia: origins and clinical relevance. J Nutr 127:990S–991S
Williams GR, Rier HN, McDonald A et al (2019) Sarcopenia & aging in cancer. J Geriatr Oncol 10:374–377
Bozzetti F (2020) Chemotherapy-induced sarcopenia. Curr Treat Options Oncol 21:7
Feliciano EC, Lee V, Chen W, Prado C, Shachar SS, Alexeeff S, Caan BJ (2019) Adiposity, muscle mass and delays and dose reduction on adjuvant taxane based chemotherapy for breast cancer. AACR2019. https://doi.org/10.1158/1538-7445.AM2019-3284
Cespedes Feliciano EM, Chen WY, Bradshaw PT et al (2019) Adipose tissue distribution and cardiovascular disease risk among breast cancer survivors. J Clin Oncol 37:2528
Hamrick MW, McGee-Lawrence ME, Frechette DM (2016) Fatty infiltration of skeletal muscle: mechanisms and comparisons with bone marrow adiposity. Front Endocrinol (Lausanne) 7:69
Martin L, Birdsell L, Macdonald N et al (2013) Cancer cachexia in the age of obesity: skeletal muscle depletion is a powerful prognostic factor, independent of body mass index. J Clin Oncol 31:1539–1547
Williams GR, Deal AM, Muss HB et al (2018) Frailty and skeletal muscle in older adults with cancer. J Geriatr Oncol 9:68–73
Williams GR, Deal AM, Muss HB et al (2017) Skeletal muscle measures and physical function in older adults with cancer: sarcopenia or myopenia? Oncotarget 8:33658–33665
Rier HN, Jager A, Sleijfer S et al (2017) Low muscle attenuation is a prognostic factor for survival in metastatic breast cancer patients treated with first line palliative chemotherapy. Breast 31:9–15
Aleixo GFP, Shachar SS, Nyrop KA et al (2020) Myosteatosis and prognosis in cancer: systematic review and meta-analysis. Crit Rev Oncol Hematol 145:102839
Shachar SS, Deal AM, Weinberg M et al (2017) Body composition as a predictor of toxicity in patients receiving anthracycline and taxane-based chemotherapy for early-stage breast cancer. Clin Cancer Res 23:3537–3543
Prado CM, Baracos VE, McCargar LJ et al (2009) Sarcopenia as a determinant of chemotherapy toxicity and time to tumor progression in metastatic breast cancer patients receiving capecitabine treatment. Clin Cancer Res 15:2920–2926
Kroenke CH, Prado CM, Meyerhardt JA et al (2018) Muscle radiodensity and mortality in patients with colorectal cancer. Cancer 124:3008–3015
Deluche E, Leobon S, Desport JC et al (2018) Impact of body composition on outcome in patients with early breast cancer. Support Care Cancer 26:861–868
Almasaudi AS, Dolan RD, McSorley ST et al (2019) Relationship between computed tomography-derived body composition, sex, and post-operative complications in patients with colorectal cancer. Eur J Clin Nutr 73:1450–1457
Nam SY, Kim BC, Han KS et al (2010) Abdominal visceral adipose tissue predicts risk of colorectal adenoma in both sexes. Clin Gastroenterol Hepatol 8(443–50):e1–2
Pielkenrood BJ, van Urk PR, van der Velden JM et al (2020) Impact of body fat distribution and sarcopenia on the overall survival in patients with spinal metastases receiving radiotherapy treatment: a prospective cohort study. Acta Oncol 59:291–297
Kaneko G, Miyajima A, Yuge K et al (2015) Visceral obesity is associated with better recurrence-free survival after curative surgery for Japanese patients with localized clear cell renal cell carcinoma. Jpn J Clin Oncol 45:210–216
Iwase T, Sangai T, Fujimoto H et al (2020) Quality and quantity of visceral fat tissue are associated with insulin resistance and survival outcomes after chemotherapy in patients with breast cancer. Breast Cancer Res Treat 179:435–443
Acknowledgements
We greatly appreciate the active support of oncology clinicians and their research staff at multiple sites and, most importantly, the breast cancer patients participating in our study. We thank Tucker Brenizer, Shanah R. Kirk, and Amy Garrett for their commitment to study implementation best practices.
Funding
This study was supported by the Breast Cancer Research Foundation (New York), UNC Lineberger Comprehensive Cancer Center/University Cancer Research Fund, and Kay Yow Fund (Raleigh North Carolina).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Gabriel FP Aleixo, Allison M Deal, Kirsten A. Nyrop, Hyman B Muss, Yi Tang Chen, Grant R Williams, Hyeon Yu, and Shlomit S Shachar declare they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
This article does not contain any animals performed by any of the authors. Informed consent was obtained from all individuals participants in this study and approved by the IRB of the University of North Carolina at Chapel Hill.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Aleixo, G.F.P., Shachar, S.S., Deal, A.M. et al. The association of body composition parameters and adverse events in women receiving chemotherapy for early breast cancer. Breast Cancer Res Treat 182, 631–642 (2020). https://doi.org/10.1007/s10549-020-05731-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-020-05731-1