Abstract
Background
Emerging preclinical evidence indicates statins, medications commonly used in the prevention of cardiovascular disease (CVD), inhibit proliferation, promote apoptosis and limit invasiveness of esophageal adenocarcinoma (EAC). Population-based observational data demonstrate statin treatment after diagnosis of EAC is associated with significant reductions in all-cause and cancer-specific mortality. A feasibility study of adjuvant statin therapy following potentially curative resection for EAC has been completed, with planned progression to a full phase III, randomized controlled trial.
Objective
The aim was to estimate the cost-utility of statin therapy following surgical resection for EAC from a UK National Health Service (NHS) perspective.
Methods
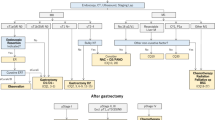
A Markov model was developed to estimate the costs and outcomes [quality-adjusted life years (QALYs)] for hypothetical cohorts of patients with EAC exposed or not exposed to statins following potentially curative surgical resection. Model parameters were based on estimates from published observational and trial data. Costs, utilities and transition probabilities were modeled to reflect clinical practice from a payer’s perspective. Probabilistic and one-way sensitivity analyses were performed to account for uncertainty in key parameters.
Results
Overall, a cost saving of £6781 per patient was realized with statin treatment compared to no statins. In probabilistic sensitivity analysis, 99% of all iterations were cost saving and 99% of all iterations were less than £20,000 per QALY gained. These results were robust to changes in the price and effectiveness of statins.
Conclusions
The cohort exposed to statins had lower costs and better QALY outcomes than the no statin cohort. Assuming a causal improvement in disease outcomes following resection for EAC, statin therapy is very likely to be a cost-saving treatment.



Similar content being viewed by others
References
Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012: Globocan 2012. Int J Cancer. 2015;136(5):E359–86.
Cancer Research UK. Number of new cases, crude and European age-standardised (AS) incidence rates per 100,000 population, UK. 2013. http://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/oesophageal-cancer. Accessed July 2016.
Lepage C, Rachet B, Jooste V, Faivre J, Coleman MP. Continuing rapid increase in esophageal adenocarcinoma in England and Wales. Am J Gastroenterol. 2008;103(11):2694–9.
Arnold M, Soerjomataram I, Ferlay J, Forman D. Global incidence of oesophageal cancer by histological subtype in 2012. Gut. 2015;64(3):381–7.
Edgren G, Adami H-O, Vainio EW, Nyren O. A global assessment of the oesophageal adenocarcinoma epidemic. Gut. 2013;62(10):1406–14.
National Audit Office. Progress in improving stroke care: report on the findings from our modelling of stroke care provision. February 2010.
van Hagen P, Hulshof MCCM, van Lanschot JJB, Steyerberg EW, van Henegouwen MIB, Wijnhoven BPL, Richel DJ, Nieuwenhuijzen GAP, Hospers GAP, Bonenkamp JJ, Cuesta MA, Blaisse RJB, Busch ORC, ten Kate FJW, Creemers G-J, Punt CJA, Plukker JTM, Verheul HMW, Bilgen EJS, van Dekken H, van der Sangen MJC, Rozema T, Biermann K, Beukema JC, Piet AHM, van Rij CM, Reinders JG, Tilanus HW, van der Gaast A. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med. 2012;366(22):2074–84.
NICE guidelines CG181. Cardiovascular disease: risk assessment and reduction, including lipid modification. July 2014. https://www.nice.org.uk/guidance/cg181/resources/cardiovascular-disease-risk-assessment-and-reduction-including-lipid-modification-pdf-35109807660997. Accessed Nov 2016.
Ogunwobi OO, Beales ILP. Statins inhibit proliferation and induce apoptosis in Barrett’s esophageal adenocarcinoma cells. Am J Gastroenterol. 2008;103(4):825–37.
Sadaria MR, et al. Statin therapy attenuates growth and malignant potential of human esophageal adenocarcinoma cells. J Thorac Cardiovasc Surg. 2011;142(5):1152–60.
Ye F. Suppression of esophageal cancer cell growth using curcumin, (-)-epigallocatechin-3-gallate and lovastatin. World J Gastroenterol. 2012;18(2):126.
Konturek PC, Burnat G, Hahn EG, et al. Inhibition of Barrett’s adenocarcinoma cell growth by simvastatin: involvement of COX-2 and apoptosis-related proteins’. J Physiol Pharmacol. 2007;58:141.
Mei Z, Liang M, Li L, Zhang Y, Wang Q, Yang W. Effects of statins on cancer mortality and progression: a systematic review and meta-analysis of 95 cohorts including 1,111,407 individuals: statins on cancer mortality and progression. Int J Cancer. 2017;140(5):1068–81.
Alexandre L, Clark AB, Bhutta HY, Chan SSM, Lewis MPN and Hart AR. Association between statin use after diagnosis of esophageal cancer and survival: a population-based cohort study. Gastroenterology. 2016;150(4):854–65.
Alexandre L, et al. PWE-109 A feasibility study of adjuvant statin therapy in the prevention of post-operative recurrence of oesophageal adenocarcinoma (The STAT-ROC Feasibility Study). London: BMJ Publishing Group; 2017.
Chadwick G, Varagunam M, Brand C, Cromwell D, Maynard N, Riley S, Crosby T, Michalowski J, National oesophago-gastric cancer audit 2016. Healthcare Quality Improvement Partnership Ltd. (HQIP) 2016.
NICE guidelines. Guide to the methods of technology appraisal 2013: Process and methods guides. (NICE article [PMG9]). https://www.nice.org.uk/process/pmg9/chapter/foreword. Accessed Nov 2016.
Cunningham D, Allum WH, Stenning SP, Thompson JN, Van de Velde CJH, Nicolson M, Scarffe JH, Lofts FJ, Falk SJ, Iveson TJ, Smith DB, Langley RE, Verma M, Weeden S. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med. 2006;355(1):11–20.
Weibull W. A statistical distribution function of wide applicability. ASME J Appl Mech. 1951;18:293–7.
Akaike H. A new look at the statistical model identification. IEEE Trans Autom Control. 1974;19(6):716–23.
Komogorov A. Sulla determinazione empirica di una legge di distribuzione. G Ist Ital Attuari. 1933;4:83–91.
Armitage J. The safety of statins in clinical practice. The Lancet. 2007;370(9601):1781–90.
Finegold JA, Manisty CH, Goldacre B, Barron AJ, Francis DP. What proportion of symptomatic side effects in patients taking statins are genuinely caused by the drug? Systematic review of randomized placebo-controlled trials to aid individual patient choice. Eur J Prev Cardiol. 2014;21(4):464–74.
Cholesterol Treatment Trialists’ (CTT) Collaboration. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170 000 participants in 26 randomised trials. The Lancet. 2010;376(9753):1670–81.
Tiwari A, Bansal V, Chugh A, Mookhtiar K. Statins and myotoxicity: a therapeutic limitation. Expert Opin Drug Saf. 2006;5(5):651–66.
Thompson PD. Statin-associated myopathy. JAMA. 2003;289(13):1681.
Thompson PD, Panza G, Zaleski A, Taylor B. Statin-associated side effects. J Am Coll Cardiol. 2016;67(20):2395–410.
Clarke P, Gray A, Legood R, Briggs A, Holman R. The impact of diabetes-related complications on healthcare costs: results from the United Kingdom Prospective Diabetes Study (UKPDS Study No. 65). Diabet Med. 2003;206:442–50.
World Health Organization. 2008–2013 action plan for the global strategy for the prevention and control of noncommunicable diseases. 2008. http://www.who.int/nmh/publications/ncd_action_plan_en.pdf. Accessed Nov 2016.
Office for National Statistics. National life tables, Great Britain, based on data for the years 2011–2013. 2014. https://www.ons.gov.uk/peoplepopulationandcommunity/birthsdeathsandmarriages/lifeexpectancies/bulletins/nationallifetablesunitedkingdom/2014-09-25. Accessed July 2016.
Saba NF, El-Rayes BF, editors. Esophageal cancer. Cham: Springer International Publishing; 2015.
Curtis L. Unit costs of health and social care. Canterbury: University of Kent; 2016.
Joint Formulary Committee. British National Formulary (online). London: BMJ Group and Pharmaceutical Press; 2016.
NHS reference costs 2014-15. https://www.gov.uk/government/publications/nhs-reference-costs-2014-to-2015. Accessed July 2017.
NICE Technology guidance [TA191]: Capecitabine for the treatment of advanced gastric cancer. July 2010. https://www.nice.org.uk/guidance/ta191. Accessed July 2016.
Chadwick G, Groene O, Cromwell D, Hardwick R, Riley S, Crosby T, Greenaway K. The Royal College of Surgeons of England, National oesophago-gastric cancer audit 2013. http://www.hscic.gov.uk/catalogue/PUB11093/clin-audi-supp-prog-oeso-gast-2013-rep.pdf. Accessed July 2016.
Thomas K, Corner H, Stobbart-Rowlands M. National primary care audit in end of life care and ACP and recommendations for improvement. BMJ Support Palliat Care. 2012;2(2):192.
Hex N, Bartlett C, Wright D, Taylor M, Varley D. Estimating the current and future costs of type 1 and type 2 diabetes in the UK, including direct health costs and indirect societal and productivity costs: estimating current and future costs of type 1 and type 2 diabetes in the UK. Diabet Med. 2012;29(7):855–62.
Liebl A, Khunti K, Orozco-Beltran D, Yale J-F. Health economic evaluation of type 2 diabetes mellitus: a clinical practice focused review. Clin Med Insights Endocrinol Diabetes. 2015;8:13.
Bowrey DJ, et al. A randomised controlled trial of six weeks of home enteral nutrition versus standard care after oesophagectomy or total gastrectomy for cancer: report on a pilot and feasibility study. Trials. 2015;16(1):531.
de Boer AGEM, et al. Transhiatal vs extended transthoracic resection in oesophageal carcinoma: patients’ utilities and treatment preferences. Br J Cancer. 2002;86(6):851–7.
Donnan P et al. Development of a decision support tool for primary care management of patients with abnormal liver function tests without clinically apparent liver disease: a record-linkage population cohort study and decision analysis (ALFIE). Health Technol. Assess. 2009;13(25):156.
Beaudet A, Clegg J, Thuresson P-O, Lloyd A, McEwan P. Review of utility values for economic modeling in type 2 diabetes. Value Health. 2014;17(4):462–70.
Clarke P, Gray A, Holman R. Estimating utility values for health states of type 2 diabetic patients using the EQ-5D (UKPDS 62). Med Decis Making. 2002;22(4):340–9.
Mitchell D, Guertin J, Iliza A, LeLorier J. Economic evaluation of a pharmacogenomic test for statin-induced myopathy in cardiovascular high-risk patients initiating a statin. Value Health. 2015;18(7):A396.
Hutchison CA, Patel K, Whitehouse T. Early survival and duration of hospital admission in rhabdomyolysis: ICNARC Case Mix Programme Database. Crit Care. 2011;15(6):452.
Briggs AH, Claxton K, Sculpher MJ. Decision modelling for health economic evaluation. Oxford: Oxford University Press; 2006.
Vemer P, Corro Ramos I, van Voorn GAK, Al MJ, Feenstra TL. AdViSHE: a validation-assessment tool of health-economic models for decision makers and model users. PharmacoEconomics. 2016;34(4):349–61.
Choi SE, Perzan KE, Tramontano AC, Kong CY, Hur C. Statins and aspirin for chemoprevention in Barrett’s esophagus: results of a cost-effectiveness analysis. Cancer Prev Res (Phila Pa.). 2014;7(3):341–50.
Thorn J, Coast J, Andronis L. Interpretation of the expected value of perfect information and research recommendations: a systematic review and empirical investigation. Med Decis Making. 2016;36(3):285–95.
Law M, Rudnicka AR. Statin safety: a systematic review. Am J Cardiol. 2006;97(8):S52–60.
Author information
Authors and Affiliations
Contributions
RFSK, CS, LA, AH and ML contributed to the writing and revision of the final manuscript. CS designed the model, with RFSK and LA contributing to the collection and analysis of data.
Corresponding author
Ethics declarations
Funding
The authors did not receive any grant support.
Conflict of Interest
R.FSK, C.S., L.A., A.H. and M.L. have no conflicts of interest to declare.
Data Availability Statement
The data and model are available on request from the corresponding author.
Rights and permissions
About this article
Cite this article
Fong Soe Khioe, R., Skedgel, C., Hart, A. et al. Adjuvant Statin Therapy for Esophageal Adenocarcinoma: A Cost-Utility Analysis. PharmacoEconomics 36, 349–358 (2018). https://doi.org/10.1007/s40273-017-0594-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40273-017-0594-1