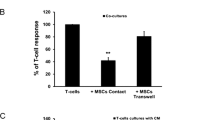
Abstract
Bone marrow-derived mesenchymal stromal cells (BM-MSCs) are multipotent progenitor cells that have shown promise for several different therapeutic applications. As they are able to modulate the function of several types of immune cells, BM-MSCs are highly important in the field of cell-based immunotherapy. Understanding BM-MSC-natural killer (NK) cell interactions is crucial for improving their therapeutic efficiency. Here, we observed that the type of NK cell-activating cytokine (e.g., IL-2, IL-12, IL-15 and IL-21) strongly influenced the outcomes of their interactions with BM-MSCs. The expression patterns of the ligands (CD112, CD155, ULPB-3) and receptors (LAIR, NCR) mediating the cross-talk between BM-MSCs and NK cells were critically modulated following co-culture. BM-MSCs partially impaired NK cell proliferation but up-regulated their secretion of IFN-γ and TNF-α. As they are cytotoxic, activated NK cells induced the killing of BM-MSCs. Indeed, BM-MSCs triggered the degranulation of NK cells and increased their release of perforin and granzymes. Interestingly, activated NK cells induced ROS generation within BM-MSCs that caused their decreased viability and reduced expression of serpin B9. Collectively, our observations reveal that BM-MSC-NK cell interactions may impact the immunobiology of both cell types. The therapeutic potential of BM-MSCs will be significantly improved once these issues are well characterized.











Similar content being viewed by others
References
Abdelrazik H, Spaggiari GM, Chiossone L, Moretta L (2011) Mesenchymal stem cells expanded in human platelet lysate display a decreased inhibitory capacity on T- and NK-cell proliferation and function. Eur J Immunol 41:3281–3290. https://doi.org/10.1002/eji.201141542
Aggarwal S, Pittenger MF (2005) Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood 105:1815–1822. https://doi.org/10.1182/blood-2004-04-1559
Almeida CR, Vasconcelos DP, Gonçalves RM, Barbosa MA (2012) Enhanced mesenchymal stromal cell recruitment via natural killer cells by incorporation of inflammatory signals in biomaterials. J R Soc Interface 9:261–271. https://doi.org/10.1098/rsif.2011.0357
Alter G, Malenfant JM, Altfeld M (2004) CD107a as a functional marker for the identification of natural killer cell activity. J Immunol Methods 294:15–22. https://doi.org/10.1016/j.jim.2004.08.008
Bartholomew A, Sturgeon C, Siatskas M et al (2002) Mesenchymal stem cells suppress lymphocyte proliferation in vitro and prolong skin graft survival in vivo. Exp Hematol 30:42–48
Biron CA, Nguyen KB, Pien GC et al (1999) Natural killer cells in antiviral defense: function and regulation by innate cytokines. Annu Rev Immunol 17:189–220. https://doi.org/10.1146/annurev.immunol.17.1.189
Blanco B, Herrero-Sánchez MC, Rodríguez-Serrano C et al (2016) Immunomodulatory effects of bone marrow versus adipose tissue derived mesenchymal stromal cells on NK cells: implications in the transplantation setting. Eur J Haematol 97:528–537. https://doi.org/10.1111/ejh.12765
Bottino C, Castriconi R, Pende D et al (2003) Identification of PVR (CD155) and Nectin-2 (CD112) as cell surface ligands for the human DNAM-1 (CD226) activating molecule. J Exp Med 198:557–67. https://doi.org/10.1084/jem.20030788
Bouchlaka MN, Redelman D, Murphy WJ (2010) Immunotherapy following hematopoietic stem cell transplantation: potential for synergistic effects. Immunotherapy 2:399–418. https://doi.org/10.2217/imt.10.20
Bryceson YT, March ME, Ljunggren H-G, Long EO (2006) Activation, coactivation, and costimulation of resting human natural killer cells. Immunol Rev 214:73–91. https://doi.org/10.1111/j.1600-065X.2006.00457.x
Chatterjee D, Marquardt N, Tufa DM et al (2014a) Role of gamma-secretase in human umbilical-cord derived mesenchymal stem cell mediated suppression of NK cell cytotoxicity. Cell Commun Signal 12:63. https://doi.org/10.1186/s12964-014-0063-9
Chatterjee D, Marquardt N, Tufa DM et al (2014b) Human umbilical cord-derived mesenchymal stem cells utilize Activin-a to suppress interferon-Î3 production by natural killer cells. Front Immunol 5:662. https://doi.org/10.3389/fimmu.2014.00662
Chen X, Song M, Zhang B, Zhang Y (2016) Reactive oxygen species regulate T cell immune response in the tumor microenvironment. Oxidative Med Cell Longev 2016:1–10. https://doi.org/10.1155/2016/1580967
Chieregato K, Albiero E, Castegnaro S et al (2012) A study on mutual interaction between cytokine induced killer cells and umbilical cord-derived mesenchymal cells: implication for their in-vivo use. Blood Cells Mol Dis 49:159–165. https://doi.org/10.1016/j.bcmd.2012.05.009
Crop MJ, Korevaar SS, de Kuiper R et al (2011) Human mesenchymal stem cells are susceptible to lysis by CD8(+) T cells and NK cells. Cell Transplant 20:1547–1559. https://doi.org/10.3727/096368910X564076
Cui R, Rekasi H, Hepner-Schefczyk M et al (2016) Human mesenchymal stromal/stem cells acquire immunostimulatory capacity upon cross-talk with natural killer cells and might improve the NK cell function of immunocompromised patients. Stem Cell Res Ther 7:88. https://doi.org/10.1186/s13287-016-0353-9
de Rham C, Ferrari-Lacraz S, Jendly S et al (2007) The proinflammatory cytokines IL-2, IL-15 and IL-21 modulate the repertoire of mature human natural killer cell receptors. Arthritis Res Ther 9:R125. https://doi.org/10.1186/ar2336
de Witte SFH, Merino AM, Franquesa M et al (2017) Cytokine treatment optimises the immunotherapeutic effects of umbilical cord-derived MSC for treatment of inflammatory liver disease. Stem Cell Res Ther 8:140. https://doi.org/10.1186/s13287-017-0590-6
DelaRosa O, Sánchez-Correa B, Morgado S et al (2012) Human adipose-derived stem cells impair natural killer cell function and exhibit low susceptibility to natural killer-mediated lysis. Stem Cells Dev 21:1333–1343. https://doi.org/10.1089/scd.2011.0139
Dominici M, Le Blanc K, Mueller I et al (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 8:315–317. https://doi.org/10.1080/14653240600855905
Dunavin N, Dias A, Li M, McGuirk J (2017) Mesenchymal stromal cells: what is the mechanism in acute graft-versus-host disease? Biomedicine 5:39. https://doi.org/10.3390/biomedicines5030039
El Haddad N, Moore R, Heathcote D et al (2011) The novel role of SERPINB9 in cytotoxic protection of human mesenchymal stem cells. J Immunol 187:2252–2260. https://doi.org/10.4049/jimmunol.1003981
Enk J, Mandelboim O (2014) The role of natural cytotoxicity receptors in various pathologies: emphasis on type I diabetes. Front Immunol 5:4. https://doi.org/10.3389/fimmu.2014.00004
Friedenstein AJ, Chailakhyan RK, Latsinik NV et al (1974a) Stromal cells responsible for transferring the microenvironment of the hemopoietic tissues. Cloning in vitro and retransplantation in vivo Transplantation 17:331–340
Friedenstein AJ, Deriglasova UF, Kulagina NN et al (1974b) Precursors for fibroblasts in different populations of hematopoietic cells as detected by the in vitro colony assay method. Exp Hematol 2:83–92
Fu Q, Man X, Yu M et al (2017) Human decidua mesenchymal stem cells regulate decidual natural killer cell function via interactions between collagen and leukocyte-associated immunoglobulin-like receptor 1. Mol Med Rep 16:2791–2798. https://doi.org/10.3892/mmr.2017.6921
Giuliani M, Bennaceur-Griscelli A, Nanbakhsh A et al (2014) TLR ligands stimulation protects MSC from NK killing. Stem Cells 32:290–300. https://doi.org/10.1002/stem.1563
Giuliani M, Oudrhiri N (2011) Human mesenchymal stem cells derived from induced pluripotent stem cells down-regulate NK-cell cytolytic machinery. Blood 118:3254–3262. https://doi.org/10.1182/blood-2010-12-325324
Götherström C, Lundqvist A, Duprez IR et al (2011) Fetal and adult multipotent mesenchymal stromal cells are killed by different pathways. Cytotherapy 13:269–278. https://doi.org/10.3109/14653249.2010.523077
He Y, Tian Z (2016) NK cell education via nonclassical MHC and non-MHC ligands. Cell Mol Immunol. https://doi.org/10.1038/cmi.2016.26
Hoogduijn MJ, Roemeling-van Rhijn M, Korevaar SS et al (2011) Immunological aspects of allogeneic and autologous mesenchymal stem cell therapies. Hum Gene Ther 22:1587–1591. https://doi.org/10.1089/hum.2011.039
Jacquemin G, Margiotta D, Kasahara A et al (2015) Granzyme B-induced mitochondrial ROS are required for apoptosis. Cell Death Differ 22:862–874. https://doi.org/10.1038/cdd.2014.180
Jewett A, Arasteh A, Tseng HC et al (2010) Strategies to rescue mesenchymal stem cells (MSCs) and dental pulp stem cells (DPSCs) from NK cell mediated cytotoxicity. PLoS One 5:1–14. https://doi.org/10.1371/journal.pone.0009874
Joyce MG, Sun PD (2011) The structural basis of ligand recognition by natural killer cell receptors. J Biomed Biotechnol 2011:203628. https://doi.org/10.1155/2011/203628
Kaiserman D, Bird PI (2010) Control of granzymes by serpins. Cell Death Differ 17:586–595. https://doi.org/10.1038/cdd.2009.169
Krampera M, Galipeau J, Shi Y et al (2013) Immunological characterization of multipotent mesenchymal stromal cells-the international society for cellular therapy (ISCT) working proposal. Cytotherapy 15:1054–1061. https://doi.org/10.1016/j.jcyt.2013.02.010
Krzewski K, Coligan JE (2012) Human NK cell lytic granules and regulation of their exocytosis. Front Immunol. https://doi.org/10.3389/fimmu.2012.00335
Lebbink RJ, van den BMCW, de RT et al (2008) The soluble leukocyte-associated Ig-like receptor (LAIR)-2 antagonizes the collagen/LAIR-1 inhibitory immune interaction. J Immunol 180:1662–1669. https://doi.org/10.4049/jimmunol.180.3.1662
Li Y, Qu Y, Wu Y et al (2011) Bone marrow mesenchymal stem cells reduce the antitumor activity of cytokine-induced killer/natural killer cells in K562 NOD/SCID mice. Ann Hematol 90:873–885. https://doi.org/10.1007/s00277-011-1156-9
Lupatov AY, Kim YS, Bystrykh OA et al (2017) Effect of fibroblast-like cells of mesenchymal origin of cytotoxic activity of lymphocytes against NK-sensitive target cells. Bull Exp Biol Med 162:552–557. https://doi.org/10.1007/s10517-017-3658-5
Mangan MSJ, Bird CH, Kaiserman D et al (2016) A novel serpin regulatory mechanism. J Biol Chem 291:3626–3638. https://doi.org/10.1074/jbc.M115.699298
Moretta A, Bottino C, Vitale M et al (2001) Activating receptors and coreceptors involved in human natural killer cell-mediated cytolysis. Annu Rev Immunol 19:197–223. https://doi.org/10.1146/annurev.immunol.19.1.197
Moretta L, Moretta A (2004) Unravelling natural killer cell function: triggering and inhibitory human NK receptors. EMBO J 23:255–259. https://doi.org/10.1038/sj.emboj.7600019
Najar M, Rouas R, Raicevic G et al (2009) Mesenchymal stromal cells promote or suppress the proliferation of T lymphocytes from cord blood and peripheral blood: the importance of low cell ratio and role of interleukin-6. Cytotherapy 11:570–583. https://doi.org/10.1080/14653240903079377
Noone C, Kihm A, English K et al (2013) IFN-γ stimulated human umbilical-tissue-derived cells potently suppress NK activation and resist NK-mediated cytotoxicity in vitro. Stem Cells Dev 22:3003–3014. https://doi.org/10.1089/scd.2013.0028
Padgett LE, Broniowska KA, Hansen PA et al (2013) The role of reactive oxygen species and proinflammatory cytokines in type 1 diabetes pathogenesis. Ann N Y Acad Sci 1281:16–35. https://doi.org/10.1111/j.1749-6632.2012.06826.x
Panetta NJ, Gupta DM, Quarto N, Longaker MT (2009) Mesenchymal cells for skeletal tissue engineering. Panminerva Med 51:25–41
Pazina T, Shemesh A, Brusilovsky M et al (2017) Regulation of the functions of natural cytotoxicity receptors by interactions with diverse ligands and alterations in splice variant expression. Front Immunol 8:369. https://doi.org/10.3389/fimmu.2017.00369
Perussia (1996) The cytokine profile of resting and activated NK cells. Methods 9:370–378
Poggi A, Prevosto C, Massaro A-M et al (2005) Interaction between human NK cells and bone marrow stromal cells induces NK cell triggering: role of NKp30 and NKG2D receptors. J Immunol 175:6352–6360. https://doi.org/10.4049/jimmunol.175.10.6352
Poggi A, Zocchi MR (2014) NK cell autoreactivity and autoimmune diseases. Front Immunol 5:27. https://doi.org/10.3389/fimmu.2014.00027
Pradier A, Passweg J, Villard J, Kindler V (2011) Human bone marrow stromal cells and skin fibroblasts inhibit natural killer cell proliferation and cytotoxic activity. Cell Transplant 20:681–691. https://doi.org/10.3727/096368910X536545
Prockop DJ (1997) Marrow stromal cells as stem cells for nonhematopoietic tissues. Science 276:71–74
Rasmusson I, Ringdén O, Sundberg B, Le Blanc K (2003) Mesenchymal stem cells inhibit the formation of cytotoxic T lymphocytes, but not activated cytotoxic T lymphocytes or natural killer cells. Transplantation 76:1208–1213. https://doi.org/10.1097/01.TP.0000082540.43730.80
Ribeiro A, Laranjeira P, Mendes S et al (2013) Mesenchymal stem cells from umbilical cord matrix, adipose tissue and bone marrow exhibit different capability to suppress peripheral blood B, natural killer and T cells. Stem Cell Res Ther 4:125–141. https://doi.org/10.1186/scrt336
Romee R, Leong JW, Fehniger TA (2014) Utilizing cytokines to function-enable human NK cells for the immunotherapy of cancer. Scientifica (Cairo) 2014:205796. https://doi.org/10.1155/2014/205796
Solana R, Casado JG, Delgado E et al (2007) Lymphocyte activation in response to melanoma: interaction of NK-associated receptors and their ligands. Cancer Immunol Immunother 56:101–109. https://doi.org/10.1007/s00262-006-0141-y
Sotiropoulou PA, Perez SA, Gritzapis AD et al (2006) Interactions between human mesenchymal stem cells and natural killer cells. Stem Cells 24:74–85. https://doi.org/10.1634/stemcells.2004-0359
Spaggiari GM, Capobianco A, Abdelrazik H et al (2008) Mesenchymal stem cells inhibit natural killer-cell proliferation, cytotoxicity, and cytokine production: role of indoleamine 2,3-dioxygenase and prostaglandin E2. Blood 111:1327–1333. https://doi.org/10.1182/blood-2007-02-074997
Spaggiari GM, Capobianco A, Becchetti S et al (2006) Mesenchymal stem cell – natural killer cell interactions: evidence that activated NK cells are capable of killing MSCs, whereas MSCs can inhibit IL-2 – induced NK-cell proliferation. Blood 107:1484–1490. https://doi.org/10.1182/blood-2005-07-2775.Supported
Thomas H, Jäger M, Mauel K et al (2014) Interaction with mesenchymal stem cells provokes natural killer cells for enhanced IL-12/IL-18-induced interferon-gamma secretion. Mediators Inflamm 2014:1–11. https://doi.org/10.1155/2014/143463
Trapani JA, Smyth MJ (2002) Functional significance of the perforin/granzyme cell death pathway. Nat Rev Immunol 2:735–747. https://doi.org/10.1038/nri911
Verneris MR (2013) Natural killer cells and regulatory T cells: how to manipulate a graft for optimal GVL. Hematol Am Soc Hematol Educ Progr 2013:335–341. https://doi.org/10.1182/asheducation-2013.1.335
Voskoboinik I, Smyth MJ, Trapani JA (2006) Perforin-mediated target-cell death and immune homeostasis. Nat Rev Immunol 6:940–952. https://doi.org/10.1038/nri1983
Wagner AK, Kadri N, Snäll J et al (2017) Expression of CD226 is associated to but not required for NK cell education. Nat Commun 8:15627. https://doi.org/10.1038/ncomms15627
Warren HS (1996) NK cell proliferation and inflammation. Immunol Cell Biol 74:473–480. https://doi.org/10.1038/icb.1996.78
Williams MS, Henkart PA (2005) Do cytotoxic lymphocytes kill via reactive oxygen species? Immunity 22:272–274. https://doi.org/10.1016/j.immuni.2005.03.003
Wu J, Song Y, Bakker AB et al (1999) An activating immunoreceptor complex formed by NKG2D and DAP10. Science 285:730–732
Yoon SR, Kim T-D, Choi I (2015) Understanding of molecular mechanisms in natural killer cell therapy. Exp Mol Med 47:e141. https://doi.org/10.1038/emm.2014.114
Zhao Z-G, Cao Z, Xu W et al (2012) Immune protection function of multipotent mesenchymal stromal cells: role of transforming growth factor-β1. Cancer Investig 30:646–656. https://doi.org/10.3109/07357907.2012.721038
Acknowledgements
This project was supported by “Le Fonds National de la Recherche Scientifique, F.R.S.-FNRS” and the “Télévie”.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the ethics committee of the “Institut Jules Bordet” (Belgium) and with the 1964 Helsinki declaration and its later amendments.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
All authors declare that they have no conflict of interest
Additional information
Hussein Fayyad-Kazan and Laurence Lagneaux are senior co-authors.
Electronic supplementary material
ESM 1
(DOCX 15 kb)
Rights and permissions
About this article
Cite this article
Najar, M., Fayyad-Kazan, M., Meuleman, N. et al. Mesenchymal stromal cells of the bone marrow and natural killer cells: cell interactions and cross modulation. J. Cell Commun. Signal. 12, 673–688 (2018). https://doi.org/10.1007/s12079-018-0448-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12079-018-0448-4