Abstract
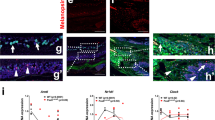
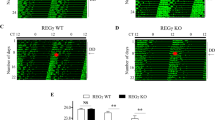
The molecular clock maintains energy constancy by producing circadian oscillations of rate-limiting enzymes involved in tissue metabolism across the day and night1,2,3. During periods of feeding, pancreatic islets secrete insulin to maintain glucose homeostasis, and although rhythmic control of insulin release is recognized to be dysregulated in humans with diabetes4, it is not known how the circadian clock may affect this process. Here we show that pancreatic islets possess self-sustained circadian gene and protein oscillations of the transcription factors CLOCK and BMAL1. The phase of oscillation of islet genes involved in growth, glucose metabolism and insulin signalling is delayed in circadian mutant mice, and both Clock5,6 and Bmal17 (also called Arntl) mutants show impaired glucose tolerance, reduced insulin secretion and defects in size and proliferation of pancreatic islets that worsen with age. Clock disruption leads to transcriptome-wide alterations in the expression of islet genes involved in growth, survival and synaptic vesicle assembly. Notably, conditional ablation of the pancreatic clock causes diabetes mellitus due to defective β-cell function at the very latest stage of stimulus–secretion coupling. These results demonstrate a role for the β-cell clock in coordinating insulin secretion with the sleep–wake cycle, and reveal that ablation of the pancreatic clock can trigger the onset of diabetes mellitus.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Green, C. B., Takahashi, J. S. & Bass, J. The meter of metabolism. Cell 134, 728–742 (2008)
Panda, S. et al. Coordinated transcription of key pathways in the mouse by the circadian clock. Cell 109, 307–320 (2002)
Rutter, J., Reick, M. & McKnight, S. L. Metabolism and the control of circadian rhythms. Annu. Rev. Biochem. 71, 307–331 (2002)
Polonsky, K. S. et al. Abnormal patterns of insulin secretion in non-insulin-dependent diabetes mellitus. N. Engl. J. Med. 318, 1231–1239 (1988)
King, D. P. et al. Positional cloning of the mouse circadian clock gene. Cell 89, 641–653 (1997)
Turek, F. W. et al. Obesity and metabolic syndrome in circadian Clock mutant mice. Science 308, 1043–1045 (2005)
Bunger, M. K. et al. Mop3 is an essential component of the master circadian pacemaker in mammals. Cell 103, 1009–1017 (2000)
Lowrey, P. L. & Takahashi, J. S. Mammalian circadian biology: elucidating genome-wide levels of temporal organization. Annu. Rev. Genomics Hum. Genet. 5, 407–441 (2004)
Kornmann, B. et al. System-driven and oscillator-dependent circadian transcription in mice with a conditionally active liver clock. PLoS Biol. 5, e34 (2007)
McCarthy, J. J. et al. Identification of the circadian transcriptome in adult mouse skeletal muscle. Physiol. Genomics 31, 86–95 (2007)
Storch, K. F. et al. Extensive and divergent circadian gene expression in liver and heart. Nature 417, 78–83 (2002)
Yang, X. et al. Nuclear receptor expression links the circadian clock to metabolism. Cell 126, 801–810 (2006)
Balsalobre, A., Damiola, F. & Schibler, U. A serum shock induces circadian gene expression in mammalian tissue culture cells. Cell 93, 929–937 (1998)
Yoo, S. H. et al. PERIOD2::LUCIFERASE real-time reporting of circadian dynamics reveals persistent circadian oscillations in mouse peripheral tissues. Proc. Natl Acad. Sci. USA 101, 5339–5346 (2004)
Yamazaki, S. et al. Resetting central and peripheral circadian oscillators in transgenic rats. Science 288, 682–685 (2000)
Drucker, D. J. The biology of incretin hormones. Cell Metab. 3, 153–165 (2006)
Gao, N. et al. Foxa2 controls vesicle docking and insulin secretion in mature β cells. Cell Metab. 6, 267–279 (2007)
Rulifson, I. C. et al. Wnt signaling regulates pancreatic β cell proliferation. Proc. Natl Acad. Sci. USA 104, 6247–6252 (2007)
Shih, D. Q. et al. Loss of HNF-1α function in mice leads to abnormal expression of genes involved in pancreatic islet development and metabolism. Diabetes 50, 2472–2480 (2001)
Stoffers, D. A. et al. Insulinotropic glucagon-like peptide 1 agonists stimulate expression of homeodomain protein IDX-1 and increase islet size in mouse pancreas. Diabetes 49, 741–748 (2000)
Withers, D. J. et al. Disruption of IRS-2 causes type 2 diabetes in mice. Nature 391, 900–904 (1998)
Gu, G., Dubauskaite, J. & Melton, D. A. Direct evidence for the pancreatic lineage: NGN3+ cells are islet progenitors and are distinct from duct progenitors. Development 129, 2447–2457 (2002)
Westgate, E. J. et al. Genetic components of the circadian clock regulate thrombogenesis in vivo . Circulation 117, 2087–2095 (2008)
Lamia, K. A., Storch, K. F. & Weitz, C. J. Physiological significance of a peripheral tissue circadian clock. Proc. Natl Acad. Sci. USA 105, 15172–15177 (2008)
Rudic, R. D. et al. BMAL1 and CLOCK, two essential components of the circadian clock, are involved in glucose homeostasis. PLoS Biol. 2, e377 (2004)
Duez, H. et al. Regulation of bile acid synthesis by the nuclear receptor Rev-erbα. Gastroenterology 135, 689–698 (2008)
Noshiro, M. et al. Multiple mechanisms regulate circadian expression of the gene for cholesterol 7α-hydroxylase (Cyp7a), a key enzyme in hepatic bile acid biosynthesis. J. Biol. Rhythms 22, 299–311 (2007)
Kudo, T., Kawashima, M., Tamagawa, T. & Shibata, S. Clock mutation facilitates accumulation of cholesterol in the liver of mice fed a cholesterol and/or cholic acid diet. Am. J. Physiol. Endocrinol. Metab. 294, E120–E130 (2008)
Fu, L. et al. The molecular clock mediates leptin-regulated bone formation. Cell 122, 803–815 (2005)
Matsuo, T. et al. Control mechanism of the circadian clock for timing of cell division in vivo . Science 302, 255–259 (2003)
Acknowledgements
We thank F. Turek, R. Allada and G. Bell for discussions and comments on the manuscript. We thank A. Kohsaka, E. Chen, J. Doering and C. Radosevich for their technical support, as well as the Biological Imaging Facility at Northwestern University and the Islet Biology Core of the University of Chicago DRTC. We thank D. Melton for the PdxCre mice. Work was supported by grants from the National Institute of Diabetes and Digestive and Kidney Diseases to K.M.R. and L.H.P.; the National Institutes of Health, Chicago Biomedical Consortium Searle Funds, and Juvenile Diabetes Research Foundation to J.B.; grant R37-ES-005703 from the National Institutes of Health to C.A.B.; and the National Institute of Mental Health to J.S.T.
Author information
Authors and Affiliations
Contributions
B.M. performed and analysed most of the experiments in this study, with technical assistance from Y.K., G.I., S.M. and C.O. E.D.B. and C.H.K. conducted and analysed real-time bioluminescence imaging experiments in isolated pancreatic islets. H.S. conducted immunostaining experiments. M.H.V. performed statistical analysis. J.P.L. conducted and analysed Ca2+ influx experiments. S.D.C. and L.J. performed statistical and gene ontogeny analysis of microarray data. C.A.B. provided Bmal1flx/flx mice. J.S.T., L.H.P., X.W., K.M.R., B.M. and J.B. provided critical intellectual input in the preparation of the manuscript. K.M.R., B.M., J.S.T. and J.B. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
[Competing Interests: J.S.T. is an Investigator in the Howard Hughes Medical Institute and a co-founder of ReSet Therapeutics Inc., and J.S.T. and J.B. are members of its scientific advisory board. J.B. is also an advisor and receives support from Amylin Pharmaceuticals.]
Supplementary information
Supplementary Information
This file contains Supplementary Methods, Supplementary Descriptions 1-5, Supplementary Figures S1-S9 with legends, Supplementary Tables S1-S3 and References. (PDF 2403 kb)
Supplementary Movie 1
Cell autonomous oscillator in pancreas. (MOV 8408 kb)
Continuous videomicroscopy monitoring of islet bioluminescence over 72 hours. Islets from Per2Luc mice were isolated via collagenase digestion and imaged as described in Supplementary Methods. The 72-hour epoch is continuously replayed.
Supplementary Movie 2
Cell autonomous oscillator in pancreas. (MOV 8397 kb)
Islets were harvested from a separate group of Per2Luc mice and monitored under identical conditions for 72 hours. The 72-hour epoch is continuously replayed.
Rights and permissions
About this article
Cite this article
Marcheva, B., Ramsey, K., Buhr, E. et al. Disruption of the clock components CLOCK and BMAL1 leads to hypoinsulinaemia and diabetes. Nature 466, 627–631 (2010). https://doi.org/10.1038/nature09253
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature09253
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.