Abstract
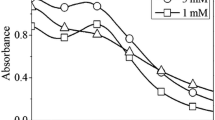
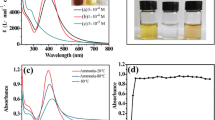
Formation of silver nanoparticles in chemical reduction of AgNO3 with peat fulvic acids in an aqueous alkali solution was studied spectrophotometrically. The size of the resulting nanoparticles and their stability were examined in relation to the reaction temperature and time, and concentrations of fulvic acids and silver nitrate.
Similar content being viewed by others
References
Suzdalev, I.P., Nanotekhnologiya: Fizikokhimiya nanoklasterov, nanostruktur i nanomaterialov (Nanotechnology: Physical Chemistry of Nanoclusters, Nanostructures, and Nanomaterials), Moscow: Kom-Kniga, 2006.
Blagitko, E.M., Burmistrov, V.A., Kolesnikov, A.P., and Mikhailov, Yu.I., Serebro v meditsine (Silver in Medicine), Novosibirsk: Nauka-Tsents, 2004.
Rao, C.N., Muller, A., and Cheetham, A.K., The Chemistry of Nanomaterials, Darmstadt: Wiley, 2004.
Pomogailo, A.D., Rozenberg, A.S., and Uflyand, I.E., Nanochastitsy metallov v polimerakh (Metal Nanoparticles in Polymers), Moscow: Khimiya, 2000.
Sviridov, V.V., Vorob’eva, T.N., Gaevskaya, T.V., and Stepanova, L.I., Khimicheskoe osazhdenie metallov v vodnykh rastvorakh (Chemical Precipitation of Metals in Aqueous Solutions), Minsk: Universitetskoe, 1987.
Loginov, A.V., Gorbunova, V.V., and Boitsova, T.B., Zh. Obshch. Khim., 1997, vol. 67, p. 189.
Kamat, P.V., Flumiani, M., and Hartland, G.V., J. Phys. Chem., 1998, vol. 102, p. 3123.
Grishchenko, L.A., Medvedeva, S.A., Aleksandrova, G.P., et al., Zh. Obshch. Khim., 2006, vol. 76, no. 7, p. 1159.
Egorova, E.M., Revina, A.A., Rostovshchikova, T.N., and Kiseleva, O.I., Vestn. Mosk. Gos. Univ., Ser. 2: Khim., 2001, vol. 42, no. 5, p. 332.
Naumova, G.V., Torf v biotekhnologii (Peat in Biotechnology), Minsk: Nauka i Tekhnika, 1987.
Orlov, D.S., Gumusovye kisloty pochv i obshchaya teoriya gumifikatsii (Humus Acids of Soils and General Theory of Humification), Moscow: Mosk. Gos. Univ., 1990.
Ponomareva, V.V. and Plotnikova, T.A., Gumus i pochvoobrazovanie (Humus and Soil Formation), Leningrad: Nauka, 1980.
Stevenson, F.J., Humic Chemistry: Genesis, Composition, Reactions, New York: Wiley, 1982, Vol. 1.
Struyk, Z. and Sposito, G., Geoderma, 2001, vol. 102, p. 329.
Matthiessen, A., J. Anal. Chem., 1996, vol. 354, p. 747.
Kefeli, V.I., Guminovye veshchestva v biosfere (Humic Substances in Biosphere), Moscow: Nauka, 1993, p. 80.
Lehmann, R., Cheng, H., and Harsh, J., Soil Sci. Soc. Am. J., 1987, vol. 51, no. 2, p. 352.
Ershov, B.G., Zh. Ross, Khim. O-va im. D.I. Mendeleeva, 2001, vol. 32, no. 3, p. 20.
Ershov, B.G., Janata, E., Henglein, A., and Fojtik A., J. Phys. Chem., 1993, vol. 97, no. 18, p. 4589.
Bohren, C.F. and Huffman, D.R., Absorption and Scattering of Light by Small Particles, New York: Wiley, 1983.
Kreibig, U. and Vollmer, M., Properties of Metal Clusters, Berlin: Springer, 1995.
Creighton, J.A. and Eadont, D.G., J. Chem. Soc. Faraday Trans., 1991, vol. 84, no. 24, p. 3881.
Ershov, B.G., Janata, E., and Henglein, A., J. Phys. Chem., 1993, vol. 97, no. 2, p. 339.
Henglein, A., J. Phys. Chem., 1993, vol. 97, no. 21, p. 5457.
Perminova, I.V., Doctoral Sci. (Chem.) Dissertation, Moscow, 2000.
Author information
Authors and Affiliations
Additional information
Original Russian Text © D.S. Sal’nikov, A.S. Pogorelova, S.V. Makarov, I.Yu. Vashurina, 2009, published in Zhurnal Prikladnoi Khimii, 2009, Vol. 82, No. 4, pp. 552–555.
Rights and permissions
About this article
Cite this article
Sal’nikov, D.S., Pogorelova, A.S., Makarov, S.V. et al. Silver ion reduction with peat fulvic acids. Russ J Appl Chem 82, 545–548 (2009). https://doi.org/10.1134/S107042720904003X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S107042720904003X