Abstract
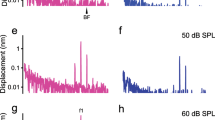
Hair cells detect vibrations of their stereociliary bundle by activation of mechanically sensitive transducer channels. Although evidence suggests the transducer channels are near the stereociliary tops and are opened by force imparted by tip links connecting contiguous stereocilia, the exact channel site remains controversial. We used fast confocal imaging of fluorescence changes reflecting calcium entry during bundle stimulation to localize the channels. Calcium signals were visible in single stereocilia of rat cochlear inner hair cells and were up to tenfold larger and faster in the second and third stereociliary rows than in the tallest first row. The number of functional stereocilia was proportional to transducer current amplitude, indicating that there were about two channels per stereocilium. Comparable results were obtained in outer hair cells. The observations, supported by theoretical simulations, suggest there are no functional mechanically sensitive transducer channels in first row stereocilia and imply the channels are present only at the bottom of the tip links.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
12 April 2009
In the version of this article initially published online, the name of the funding agency was spelt incorrectly in the acknowledgements section. The error has been corrected for the print, PDF and HTML versions of this article.
References
Fettiplace, R. & Ricci, A.J. Mechanoelectrical transduction in auditory hair cells. in Springer Handbook of Auditory Research: Hair Cells (eds. Eatock, R.A., Popper, A. & Fay, R.R.) 154–203 (Springer, Germany, 2006).
Hudspeth, A.J. Extracellular current flow and the site of transduction by vertebrate hair cells. J. Neurosci. 2, 1–10 (1982).
Jaramillo, F. & Hudspeth, A.J. Localization of the hair cell's transduction channels at the hair bundle's top by iontophoretic application of a channel blocker. Neuron 7, 409–420 (1991).
Lumpkin, E.A. & Hudspeth, A.J. Detection of Ca2+ entry through mechanosensitive channels localizes the site of mechanoelectrical transduction in hair cells. Proc. Natl. Acad. Sci. USA 92, 10297–10301 (1995).
Denk, W., Holt, J.R., Shepherd, G.M. & Corey, D.P. Calcium imaging of single stereocilia in hair cells: localization of transduction channels at both ends of tip links. Neuron 15, 1311–1321 (1995).
Pickles, J.O., Comis, S.D. & Osborne, M.P. Cross-links between stereocilia in the guinea pig organ of Corti, and their possible relation to sensory transduction. Hear. Res. 15, 103–112 (1984).
Howard, J. & Hudspeth, A.J. Compliance of the hair bundle associated with gating of mechanoelectrical transduction channels in the bullfrog's saccular hair cell. Neuron 1, 189–199 (1988).
Assad, J.A., Shepherd, G.M. & Corey, D.P. Tip-link integrity and mechanical transduction in vertebrate hair cells. Neuron 7, 985–994 (1991).
Kazmierczak, P. et al. Cadherin 23 and protocadherin 15 interact to form tip-link filaments in sensory hair cells. Nature 449, 87–91 (2007).
Ohmori, H. Mechano-electrical transduction currents in isolated vestibular hair cells of the chick. J. Physiol. (Lond.) 359, 189–217 (1985).
Ricci, A.J., Crawford, A.C. & Fettiplace, R. Tonotopic variation in the conductance of the hair cell mechanotransducer channel. Neuron 40, 983–990 (2003).
Beurg, M., Evans, M.G., Hackney, C.M. & Fettiplace, R. A large-conductance calcium-selective mechanotransducer channel in mammalian cochlear hair cells. J. Neurosci. 26, 10992–11000 (2006).
Roth, B. & Bruns, V. Postnatal development of the rat organ of Corti. II. Hair cell receptors and their supporting elements. Anat. Embryol. (Berl.) 185, 571–581 (1992).
Furness, D.N., Richardson, G.P. & Russell, I.J. Stereociliary bundle morphology in organotypic cultures of the mouse cochlea. Hear. Res. 38, 95–109 (1989).
Waguespack, J., Salles, F.T., Kachar, B. & Ricci, A.J. Stepwise morphological and functional maturation of mechanotransduction in rat outer hair cells. J. Neurosci. 27, 13890–13902 (2007).
Harasztosi, C. & Gummer, A.W. Position of calcium entry into the outer hair cell stereocilia. Assoc. Res. Otolaryngol. Abs. 258 (2008).
Nam, J.H. & Fettiplace, R. Theoretical conditions for high-frequency hair bundle oscillations in auditory hair cells. Biophys. J. 95, 4948–4962 (2008).
Gillespie, P.G. & Cyr, J.L. Myosin-1c, the hair cell's adaptation motor. Annu. Rev. Physiol. 66, 521–545 (2004).
Stauffer, E.A. et al. Fast adaptation in vestibular hair cells requires Myosin-1c activity. Neuron 47, 541–553 (2005).
Kennedy, H.J., Evans, M.G., Crawford, A.C. & Fettiplace, R. Fast adaptation of mechanoelectrical transducer channels in mammalian cochlear hair cells. Nat. Neurosci. 6, 832–836 (2003).
Wu, Y.C., Tucker, T. & Fettiplace, R. A theoretical study of calcium microdomains in turtle hair cells. Biophys. J. 71, 2256–2275 (1996).
Wu, Y-C., Ricci, A.J. & Fettiplace, R. Two components of transducer adaptation in auditory hair cells. J. Neurophysiol. 82, 2171–2181 (1999).
Lumpkin, E.A. & Hudspeth, A.J. Regulation of free Ca2+ concentration in hair-cell stereocilia. J. Neurosci. 18, 6300–6318 (1998).
Acknowledgements
We thank Redshirt Imaging and Prairie Technologies for help in optimizing the confocal system. This work was supported by National Institute on Deafness and other Communication Disorders grants RO1 DC03896 to A.J.R. and DC01362 to R.F.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary Text and Figures
Supplementary Figure 1 (PDF 207 kb)
Supplementary Video 1
Development of the calcium fluorescence in an inner hair cell bundle during hair bundle stimulation. The hair cell was depolarized to +100 mV (top trace, membrane potential) then the hair bundle was displaced (second trace, bundle displacement). The fluorescent signal began once the cell was repolarized to −80 mV, the signal being localized to the R2 stereocilia with no change in R1 stereocilia. (AVI 1572 kb)
Supplementary Video 2
Development of the calcium fluorescence in an outer hair cell bundle during hair bundle stimulation. The outer hair cell was depolarized to +100 mV (top trace, membrane potential) then the hair bundle displaced (second trace, bundle displacement). The fluorescent signal began once the cell was repolarized to −80 mV, the signal being localized to the R2 and R3 stereocilia with no change in R1 stereocilia. (AVI 1572 kb)
Rights and permissions
About this article
Cite this article
Beurg, M., Fettiplace, R., Nam, JH. et al. Localization of inner hair cell mechanotransducer channels using high-speed calcium imaging. Nat Neurosci 12, 553–558 (2009). https://doi.org/10.1038/nn.2295
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2295
This article is cited by
-
Selective binding and transport of protocadherin 15 isoforms by stereocilia unconventional myosins in a heterologous expression system
Scientific Reports (2022)
-
Prestin-Mediated Frequency Selectivity Does not Cover Ultrahigh Frequencies in Mice
Neuroscience Bulletin (2022)
-
In situ motions of individual inner-hair-cell stereocilia from stapes stimulation in adult mice
Communications Biology (2021)
-
Single-molecule force spectroscopy reveals the dynamic strength of the hair-cell tip-link connection
Nature Communications (2021)
-
Mitochondrial calcium uniporter is essential for hearing and hair cell preservation in congenic FVB/NJ mice
Scientific Reports (2021)