Abstract
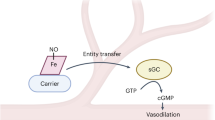
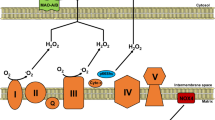
The physiological role of nitric oxide (NO) in the maintenance of vascular tone, in synaptic transmission and in cellular defence is now firmly established. Recent evidence indicates that NO can also affect mitochondrial function. Here, we review findings indicating that NO — through its interaction with components of the electron-transport chain — might function not only as a physiological regulator of cell respiration, but also to augment the generation of reactive oxygen species by mitochondria, and thereby trigger mechanisms of cell survival or death.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Wainio, W. W. Reactions of cytochrome oxidase. J. Biol. Chem. 212, 723–733 (1955).
Cleeter, M. W., Cooper, J. M., Darley-Usmar, V. M., Moncada, S. & Schapira, A. H. Reversible inhibition of cytochrome c oxidase, the terminal enzyme of the mitochondrial respiratory chain, by nitric oxide. Implications for neurodegenerative diseases. FEBS Lett. 345, 50–54 (1994).
Brown, G. C. & Cooper, C. E. Nanomolar concentrations of nitric oxide reversibly inhibit synaptosomal respiration by competing with oxygen at cytochrome oxidase. FEBS Lett. 356, 295–298 (1994).
Schweizer, M. & Richter, C. NO potently and reversibly de-energizes mitochondria at low oxygen tension. Biochem. Biophys. Res. Commun. 204, 169–175 (1994).
Torres, J., Darley-Usmar, V. & Wilson, M. T. Inhibition of cytochrome c oxidase in turnover by NO: mechanism and implications for control of respiration. Biochem. J. 312, 169–173 (1995).
Guiffre, A. et al. On the mechanism of inhibition of cytochrome c oxidase by NO. J. Biol. Chem. 271, 33404–33408 (1996).
Bellamy, T. C., Wood, J., Goodwin, D. A. & Garthwaite, J. Rapid desensitization of the nitric oxide receptor, soluble guanylyl cyclase, underlies diversity of cellular cGMP responses. Proc. Natl Acad. Sci. USA 97, 2928–2933 (2000).
Tamura, M. In vivo study of tissue oxygen metabolism using optical and nuclear magnetic resonance spectroscopies. Annu. Rev. Physiol. 51, 813–834 (1989).
Shibuki, K. & Okada, D. Endogenous nitric oxide release required for long-term synaptic depression in the cerebellum. Nature 349, 326–328 (1991).
Malinski, T. et al. Diffusion of nitric oxide in the aorta wall monitored in situ by porphyrinic microsensors. Biochem. Biophys. Res. Commun. 193, 1076–1082 (1993).
Shen, W., Hintze, T. H. & Wolin, M. S. Nitric oxide. An important signaling mechanism between vascular endothelium and parenchymal cells in the regulation of oxygen consumption. Circulation 92, 3505–3512 (1995).
Miles, P. R., Bowman, L. & Huffman, L. Nitric oxide alters metabolism in isolated alveolar type II cells. Am. J. Physiol. 271, L23–L30 (1996).
Clementi, E., Brown, G. C., Foxwell, N. & Moncada, S. On the mechanism by which vascular endothelial cells regulate their oxygen consumption. Proc. Natl Acad. Sci. USA 96, 1559–1562 (1999).
Loke, K. E. et al. Endogenous endothelial nitric oxide synthase-derived nitric oxide is a physiological regulator of myocardial oxygen consumption. Circ. Res. 84, 840–845 (1999).
Wood, J. & Garthwaite, J. Models of the diffusional spread of nitric oxide: implications for neural nitric oxide signalling and its pharmacological properties. Neuropharmacology 33, 1235–1244 (1994).
López-Figueroa, M. O. et al. Direct evidence of nitric oxide presence within mitochondria. Biochem. Biophys. Res. Commun. 272, 129–133 (2000).
Bates, T. E., Loesch, A., Burnstock, G. & Clark, J. B. Immunocytochemical evidence for a mitochondrially located nitric oxide synthase in brain and liver. Biochem. Biophys. Res. Commun. 213, 896–900 (1995).
Kobzik, L., Stringer, B., Balligand, J.-L., Reid, M. B. & Stamler, J. S. Endothelial type nitric oxide synthase in skeletal muscle fibers: mitochondrial relationships. Biochem. Biophys. Res. Commun. 211, 375–381 (1995).
Ghafourifar, P. & Richter, C. Nitric oxide synthase activity by mitochondria. FEBS Lett. 418, 291–296 (1997).
Giulivi, C., Poderoso, J. J. & Boveris, A. Production of nitric oxide by mitochondria. J. Biol. Chem. 273, 11038–11043 (1998).
Tatoyan, A. & Giulivi, C. Purification and characterization of a nitric-oxide synthase from rat liver mitochondria. J. Biol. Chem. 273, 11044–11048 (1998).
Zingarelli, B. et al. Oxidation, tyrosine nitration and cytostasis induction in the absence of inducible nitric oxide synthase. Int. J. Mol. Med. 1, 787–795 (1998).
Rothe, F., Huang, P. L. & Wolf, G. Ultrastructural localization of neuronal nitric oxide synthase in the laterodorsal tegmental nucleus of wild-type and knockout mice. Neuroscience 94, 193–201 (1999).
Jiang, B. H., Semenza, G. L., Bauer, C. & Marti, H. H. Hypoxia-inducible factor 1 levels vary exponentially over a physiologically relevant range of O2 tension. Am. J. Physiol. 271, C1172–C1180 (1996).
Chandel, N. S., Budinger, G. R. S. & Schumacker, P. T. Molecular oxygen modulates cytochrome c oxidase function. J. Biol. Chem. 271, 18672–18677 (1996).
Brand, M. D. & Murphy, M. P. Control of electron flux through the respiratory chain in mitochondria and cells. Biol. Rev. Camb. Philos. Soc. 62, 141–193 (1987).
Poderoso, J. J. et al. Nitric oxide inhibits electron transfer and increases superoxide radical production in rat heart mitochondria and submitochondrial particles. Arch. Biochem. Biophys. 28, 85–92 (1996).
Thomas, D. D., Liu, X., Kantrow, S. P. & Lancaster, J. R. Jr. The biological lifetime of nitric oxide: implications for the perivascular dynamics of NO and O2 . Proc. Natl Acad. Sci. USA 98, 355–360 (2001).
Pohl, U. & Busse, R. Hypoxia stimulates release of endothelium-derived relaxant factor. Am. J. Physiol. 256, H1595–H1600 (1989).
Trimmer, B. A. et al. Nitric oxide and the control of firefly flashing. Science 292, 2486–2488 (2001).
Rees, D. D., Monkhouse, J. E., Cambridge, D. & Moncada, S. Nitric oxide and the haemodynamic profile of endotoxin shock in the conscious mouse. Br. J. Pharmacol. 124, 540–546 (1998).
Brown, G. C., Foxwell, N. & Moncada, S. Transcellular regulation of cell respiration by nitric oxide generated by activated macrophages. FEBS Lett. 439, 321–324 (1998).
Jenkins, D. C. et al. Roles of nitric oxide in tumor growth. Proc. Natl Acad. Sci. USA 92, 4392–4396 (1995).
Haddad, J. J. & Land, S. C. A non-hypoxic, ROS-sensitive pathway mediates TNF-α-dependent regulation of HIF-1α. FEBS Lett. 505, 269–274 (2001).
Kimura, H. et al. Hypoxia response element of the human vascular endothelial growth factor gene mediates transcriptional regulation by nitric oxide: control of hypoxia-inducible factor-1 activity by nitric oxide. Blood 95, 189–197 (2000).
Palmer, L. A., Gaston, B. & Johns, R. A. Normoxic stabilization of hypoxia inducible factor 1 expression and activity: redox-dependent effect of nitrogen oxides. Mol. Pharmacol. 58, 1197–1203 (2000).
Huang, L. E., Willmore, W. G., Gu, J., Goldberg, M. A. & Bunn, H. F. Inhibition of hypoxia-inducible factor 1 activation by carbon monoxide and nitric oxide: implications for oxygen sensing and signaling. J. Biol. Chem. 274, 9038–9044 (1999).
Gryglewski, R. J., Palmer, R. M. J. & Moncada, S. Superoxide anion is involved in the breakdown of endothelium-derived vascular relaxing factor. Nature 320, 454–456 (1986).
Palmer, R. M. J., Ferrige, A. G. & Moncada, S. Nitric oxide release accounts for the biological activity of endothelium-derived relaxing factor. Nature 327, 524–526 (1987).
McCall, T. B., Boughton-Smith, N. K., Palmer, R. M. J., Whittle, B. J. R. & Moncada, S. Synthesis of nitric oxide from l-arginine by neutrophils. Biochem. J. 261, 293–296 (1989).
Beckman, J. S., Beckman, T. W., Chen, J., Marshall, P. A. & Freeman, B. A. Apparent hydroxyl radical production by peroxynitrite: implications for endothelial injury from nitric oxide and superoxide. Proc. Natl Acad. Sci. USA 87, 1620–1624 (1990).
Moro, M. A. et al. Paradoxical fate and biological action of peroxynitrite on human platelets. Proc. Natl Acad. Sci. USA 91, 6702–6706 (1994).
Lizasoain, I., Moro, M. A., Knowles, R. G., Darley-Usmar, V. & Moncada, S. Nitric oxide and peroxynitrite exert distinct effects on mitochondrial respiration which are differentially blocked by glutathione or glucose. Biochem. J. 314, 877–880 (1996).
Boveris, A. & Chance, B. The mitochondrial generation of hydrogen peroxide. Biochem. J. 134, 707–716 (1973).
Turrens, J. F. & Boveris, A. Generation of superoxide anion by the NADH dehydrogenase of bovine heart mitochondria. Biochem. J. 191, 421–427 (1980).
Boveris, A., Cadenas, E. & Stoppani, A. O. M. Role of ubiquinone in the mitochondrial generation of hydrogen peroxide. Biochem. J. 156, 435–444 (1976).
Chandel, N. S. et al. Reactive oxygen species generated at mitochondrial complex III stabilize hypoxia-inducible factor-1α during hypoxia. J. Biol. Chem. 275, 25130–25138 (2000).
Antunes, F. & Cadenas, E. Estimation of H2O2 gradients across biomembranes. FEBS Lett. 475, 121–126 (2000).
Packer, M. A., Porteous, C. M. & Murphy, M. P. Superoxide production by mitochondria in the presence of nitric oxide forms peroxynitrite. Biochem. Mol. Biol. Int. 40, 527–534 (1996).
Ischiropoulos, H. Biological tyrosine nitration: a pathophysiological function of nitric oxide and reactive oxygen species. Arch. Biochem. Biophys. 356, 1–11 (1998).
Maragos, C. M. et al. Complexes of ·NO with nucleophiles as agents for the controlled biological release of nitric oxide. Vasorelaxant effects. J. Med. Chem. 34, 3242–3247 (1991).
Clementi, E., Brown, G. C., Feelisch, M. & Moncada, S. Persistent inhibition of cell respiration by nitric oxide: crucial role of S-nitrosylation of mitochondrial complex I and protective action of glutathione. Proc. Natl Acad. Sci. USA 13, 7631–7636 (1998).
Turrens, J. F., Freeman, B. A. & Crapo, J. D. Hyperoxia increases H2O2 release by lung mitochondria and microsomes. Arch. Biochem. Biophys. 217, 411–421 (1982).
Riobo, N. A. et al. Nitric oxide inhibits mitochondrial NADH–ubiquinone reductase through peroxynitrite formation. Biochem. J. 359, 139–145 (2001).
Giulivi, C. Functional implications of nitric oxide produced by mitochondria in mitochondrial metabolism. Biochem. J. 332, 673–679 (1998).
Almeida, A. & Bolanos, J. P. A transient inhibition of mitochondrial ATP synthesis by nitric oxide synthase activation triggered apoptosis in primary cortical neurons. J. Neurochem. 77, 676–690 (2001).
Beltrán, B., Mathur, A., Duchen, M. R., Erusalimsky, J. D. & Moncada, S. The effect of nitric oxide on cell respiration: a key to understanding its role in cell survival or death. Proc. Natl Acad. Sci. USA 97, 14602–14607 (2000).
Almeida, A., Almedia, J., Bolaños, J. P. & Moncada, S. Different responses of astrocytes and neurons to nitric oxide: the role of glycolytically-generated ATP in astrocyte protection. Proc. Natl Acad. Sci. USA 98, 15294–15299 (2001).
Radi, R., Beckman, J. S., Bush, K. M. & Freeman, B. A. Peroxynitrite oxidation of sulfhydryls. The cytotoxic potential of superoxide and nitric oxide. J. Biol. Chem. 266, 4244–4250 (1991).
Bolanos, J. P. et al. Nitric oxide-mediated mitochondrial damage: a potential neuroprotective role for glutathione. Free Rad. Biol. Med. 21, 995–1001 (1996).
Beltran, B., Orsi, A., Clementi, E. & Moncada, S. Oxidative stress and S-nitrosylation of proteins in cells. Br. J. Pharmacol. 129, 953–960 (2000).
Halestrap, A. P., Woodfield, K. Y. & Connern, C. P. Oxidative stress, thiol reagents, and membrane potential modulate the mitochondrial permeability transition by affecting nucleotide binding to the adenine nucleotide translocase. J. Biol. Chem. 272, 3346–3354 (1997).
Cassina, A. M. et al. Cytochrome c nitration by peroxynitrite. J. Biol. Chem. 275, 21409–21415 (2000).
Hortelano, S., Alvarez, A. M. & Bosca, L. Nitric oxide induces tyrosine nitration and release of cytochrome c preceding an increase of mitochondrial transmembrane potential in macrophages. FASEB J. 13, 2311–2317 (1999).
Furchgott, R. F. & Zawadzki, J. V. The obligatory role of endothelial cells in the relaxation of arterial smooth muscle by acetylcholine. Nature 288, 373–376 (1980).
Palmer, R. M., Ashton, D. S. & Moncada, S. Vascular endothelial cells synthesize nitric oxide from l-arginine. Nature 333, 664–666 (1988).
Alderton, W. K., Cooper, C. E. & Knowles, R. G. Nitric oxide synthases: structure, function and inhibition. Biochem. J. 357, 593–615 (2001).
Moncada, S., Palmer, R. M. & Higgs, E. A. Nitric oxide: physiology, pathophysiology, and pharmacology. Pharmacol. Rev. 43, 109–142 (1991).
Acknowledgements
The authors wish to thank A. Higgs for her valuable contribution to this manuscript. We have drawn from an extensive literature and would like to acknowledge the considerable contributions of those colleagues whom, because of space constraints, we have not been able to quote.
Author information
Authors and Affiliations
Corresponding author
Related links
Rights and permissions
About this article
Cite this article
Moncada, S., Erusalimsky, J. Does nitric oxide modulate mitochondrial energy generation and apoptosis?. Nat Rev Mol Cell Biol 3, 214–220 (2002). https://doi.org/10.1038/nrm762
Issue Date:
DOI: https://doi.org/10.1038/nrm762
This article is cited by
-
The angiotensin-converting enzyme I/D polymorphism does not impact training-induced adaptations in exercise capacity in patients with stable coronary artery disease
Scientific Reports (2023)
-
Robust arm and leg muscle adaptation to training despite ACE inhibition: a randomized placebo-controlled trial
European Journal of Applied Physiology (2023)
-
Recent deveolpment of multifunctional responsive gas-releasing nanoplatforms for tumor therapeutic application
Nano Research (2023)
-
Shuttle between arginine and lysine: influence on cancer immunonutrition
Amino Acids (2023)
-
Inhaled nitric oxide: role in the pathophysiology of cardio-cerebrovascular and respiratory diseases
Intensive Care Medicine Experimental (2022)