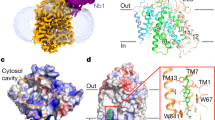
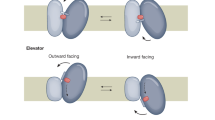
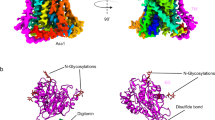
Abstract
A small group of closely related proteins is responsible for all active transport in animal cells, and inorganic cations are the only substances transported by these enzymes. They share a common kinetic mechanism in which two fundamental conformations participate, each receiving and dispatching substrates from its unique side of the membrane. During transport, the cations must pass through their enzyme to cross the membrane and intense interest is currently focused on the possibility that the path which they follow lies within the interface between two discrete subunits in a dimeric structure. Although ‘half-of-sites’ behaviour, consistent with this hypothesis, has been reported, it is now known that systematic errors were responsible for this mistaken conclusion. The number of protomers which comprise a functional unit of active transport has not been determined.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Kyte, J. J. biol. Chem. 246, 4157–4165 (1971).
MacLennan, D. H. J. biol. Chem. 245, 4508–4518 (1970).
Niggli, V., Penniston, J. T. & Carafoli, E. J. biol. Chem. 254, 9955–9958 (1979).
Sachs, G. et al. J. biol Chem. 251, 7690–7698 (1976).
Sen, A. K. & Post, R. L. J. biol. Chem. 239, 345–352 (1964).
Hasselbach, W. Biochim. biophys. Acta 515, 23–53 (1978).
Scarborough, G. A. Biochemistry 19, 2925–2931 (1980).
Dufour, J. P. & Goffeau, A. J. biol. Chem. 253, 7026–7032 (1978).
Uesugi, S. et al. J. biol. Chem. 246, 531–543 (1971).
MacLennan, D. H., Seeman, P., Iies, G. H. & Yip, C. C. J. biol Chem. 246, 2702–2710 (1971).
Saccomani, G., Shah, G., Spenney, J. G. & Sachs, G. J. biol Chem. 250, 4802–4809 (1975).
Dame, J. B. & Scarborough, G. A. Biochemistry 19, 2931–2937 (1980).
Bastide, F., Meissner, G., Fleischer, S. & Post, R. L. J. biol Chem. 248, 8385–8391 (1973).
Karlish, S. J. D., Jørgensen, P. L. & Gitler, C. Nature 269, 715–717 (1977).
Farley, R. A., Goldman, D. W. & Bayley, H. J. boil. Chem. 255, 860–864 (1980).
Allen, G., Trinnamen, B. J. & Green, N. M. Biochem. J. 187, 591–616 (1980).
Castro, J. & Farley, R. A. J. biol Chem. 254, 2221–2228 (1979).
Post, R. L. & Kume, S. J. biol Chem. 248, 6993–7000 (1973).
Albers, R. W., Fahn, S. & Koval, G. J. Proc. natn. Acad. Sci. U.S.A. 50, 474–481 (1963).
Kanazawa, T., Saito, M. & Tonomura, Y. J. Biochem. 67, 693–711 (1970).
Karlish, S. J. D., Yates, D. W. & Glynn, I. M. Biochim. biophys. Acta 525, 252–264 (1978).
Moczydlowski, E. G. & Fortes, P. A. G. J. biol. Chem. 256, 2346–2356; 2357–2366 (1981).
Karlish, S. J. D. & Yates, D. W. Biochim. biophys. Acta 527, 115–130 (1978).
Jørgensen, P. L. Biochim. biophys. Acta 401, 399–415 (1975).
Koepsell, H. J. Membrane Biol. 48, 69–94 (1979).
Fukushima, Y. & Tonomura, Y. J. Biochem. 74, 135–142 (1973).
Fahn, S., Hurley, M. R., Koval, G. J. & Albers, R. W. J. biol. Chem. 241, 1890–1895 (1966).
Fahn, S., Koval, G. J. & Albers, R. W. J. biol. Chem. 243, 1993–2002 (1968).
Mårdh, S. & Post, R. L. J. biol. Chem. 252, 633–638 (1977).
Beaugé, L. A. & Glynn, I. M. J. Physiol, Lond. 289, 17–31 (1979).
Dupont, Y. & Leigh, J. B. Nature 273, 396–398 (1978).
Takisawa, H. & Tonomura, Y. J. Biochem. 86, 425–441 (1979).
Wallmark, B., Stewart, H. B., Rabon, E., Saccomani, G. & Sachs, G. J. biol Chem. 255, 5313–5319 (1980).
Neet, K. E. & Green, N. M. Archs Biochem. Biophys. 178, 588–597 (1977).
Neufeld, A. H. & Levy, H. M. J. biol Chem. 244, 6493–6497 (1969).
Kyte, J. J. biol. Chem. 250, 7443–7449 (1975).
Bretscher, M. S. J. molec. Biol. 59, 351–357 (1971).
Singer, S. J. & Nicolson, G. N. Science 175, 720–731 (1972).
Khorana, H. G. et al. Proc. natn. Acad. Sci. U.S.A. 76, 5046–5050 (1979).
Jost, P. C., Griffith, O. H., Capaldi, R. A. & Vanderkooi, G. Proc. natn. Acad. Sci. U.S.A. 70, 480–484 (1973).
Monod, J., Wyman, J. & Changeux, J. P. J. molec. Biol. 12, 88–118 (1965).
Cullis, A. F., Muirhead, H., Perutz, M. R., Rossmann, M. G. & North, A. C. T. Proc. R. Soc. A 265, 161–187 (1962).
Craig, W. S. & Kyte, J. J. biol Chem. 255, 6262–6269 (1980).
Klingenberg, M. Nature 290, 449–454 (1981).
Kyte, J. J. biol Chem. 249, 3652–3660 (1974).
Jardetzky, O. Nature 211, 969–970 (1966).
Singer, S. J. A. Rev. Biochem. 43, 805–833 (1974).
Kyte, J. J. biol Chem. 247, 7642–7649 (1972).
Liang, S. M. & Winter, C. G. J. biol. Chem. 252, 8278–8284 (1977).
Giotta, G. J. J. biol. Chem. 251, 1247–1252 (1976).
Hastings, D. F. & Reynolds, J. A. Biochemistry 18, 817–821 (1979).
Esmann, M., Skou, J. C. & Christiansen, C. Biochim. biophys. Acta 567, 410–420 (1979).
Deguchi, N., Jørgensen, P. L. & Maunsbach, A. B. J. Cell. Biol. 75, 619–634 (1977).
Anholt, R., Lindstrom, J. & Montal, M. Eur. J. Biochem. 109, 481–487 (1980).
le Maire, M., Møller, J. V. & Tanford, C. Biochemistry 15, 2336–2342 (1976).
Schachman, H. K. & Edelstein, S. J. Biochemistry 5, 2681–2705 (1966).
Weber, K. J. biol. Chem. 243, 543–546 (1968).
Fersht, A. R. Biochemistry 14, 5–12 (1975).
Peterson, G. L., Ewing, R. D., Hootman, S. R. & Conte, F. P. J. biol Chem. 253, 4762–4770 (1978).
Jørgensen, P. L. Biochim. biophys. Acta 356, 53–67 (1974).
Jørgensen, P. L. Biochim. biophys. Acta 466, 97–108 (1977).
Perrone, J. R., Hackney, J. F., Dixon, J. F. & Hokin, L. E. J. biol. Chem. 250, 4178–4184 (1975).
Cantley, L. C. Jr, Cantley, L. G. & Josephson, L. J. biol. Chem. 253, 7361–7368 (1978).
Lowry, O. H., Rosebrough, N. J., Farr, A. L. & Randall, R. J. J. biol. Chem. 193, 265–275 (1951).
deJong, W. W., Zweers, A. & Cohen, L. H. Biochem. biophys. Res. Commun. 82, 532–539 (1978).
Rizzolo, L. J., LeMaire, M., Reynolds, J. A. & Tanford, C. Biochemistry 15, 3433–3437 (1976).
Kyte, J. J. biol. Chem. 247, 7634–7641 (1972).
Askari, A., Huang, W. & Antieau, J. M. Biochemistry 19, 1132–1140 (1980).
Meissner, G. Biochim. biophys. Acta 298, 906–926 (1973).
Steitz, T. A., Fletterick, R. J., Anderson, W. F. & Anderson, C. M. J. molec. Biol 104, 197–222 (1976).
Monteilhet, C. & Blow, D. M. J. molec. Biol. 122, 407–417 (1978).
Gennis, L. S. Proc. natn. Acad. Sci. U.S.A. 73, 3928–3932 (1976).
Scheek, R. M., Berden, J. A., Hooghiemstra, R. & Slater, E. C. Biochim. biophys. Acta 569, 124–134 (1979).
Mature, J. M. & Hollenberg, M. D. Proc. natn. Acad. Sci. U.S.A. 75, 3070–3074 (1978).
Cocivera, M., McManaman, J. & Wilson, I. B. Biochemistry 19, 2901–2907 (1980).
Rosenbusch, J. P. & Weber, K. J. biol. Chem. 246, 1644–1657 (1971).
Bonitz, S. G., Coruzzi, G., Thalenfeld, B. E., Tzagoloff, A. & Macino, G. J. biol. Chem. 255, 11927–11941 (1980).
Jørgensen, K. E., Lind, K. E., Røigaard-Petersen, H. & Møller, J. V. Biochem. J. 169, 489–498 (1978).
Dean, W. L. & Tanford, C. Biochemistry 17, 1683–1690 (1978).
Møller, J. V., Lind, K. E., & Andersen, J. P. J. biol. Chem. 255, 1912–1920 (1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kyte, J. Molecular considerations relevant to the mechanism of active transport. Nature 292, 201–204 (1981). https://doi.org/10.1038/292201a0
Published:
Issue Date:
DOI: https://doi.org/10.1038/292201a0
This article is cited by
-
Biomechanical model of the P-type ion pumps of the cell
Naturwissenschaften (1994)
-
Relationship between Potassium Concentration and Inhibitory Effects of β-Adrenergic Blockers on Myocardial Na+, K+-ATPase
Drug Investigation (1992)
-
Protein electric response signals from dielectrically polarized systems
The Journal of Membrane Biology (1989)
-
Structural basis for E1–E2 conformational transitions in Na, K-pump and Ca-pump proteins
The Journal of Membrane Biology (1988)
-
The plasma membrane ATPase ofNeurospora: A proton-pumping electroenzyme
Journal of Bioenergetics and Biomembranes (1987)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.