Abstract
Purpose
High-dose methotrexate (HD-MTX)-based chemotherapy regimen is the first-line option for primary central nervous system lymphoma (PCNSL). This prospective cohort study aimed to evaluate the efficacy and adverse effects of HD-MTX plus idarubicin (IDA) in patients with newly diagnosed immunocompetent PCNSL.
Methods
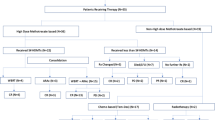
We recruited newly diagnosed PCNSL patients from January 2017 to August 2020. Patients were assigned into two groups: HD-MTX monotherapy and HD-MTX plus IDA (HD-MTX/IDA). In the HD-MTX monotherapy group, patients were treated with MTX 8 g/m2 alone on day 1, while the HD-MTX/IDA group received MTX 8 g/m2 on day 1 and IDA 10 mg/m2 on day 2. Treatments were repeated every 3 weeks for 8 cycles except for progression and/or unacceptable toxicity.
Results
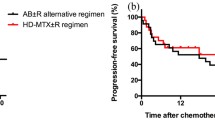
We recruited 61 PCNSL patients, including 36 in the HD-MTX and 25 in the HD-MTX/IDA group. The CR rate was 68% in the HD-MTX/IDA group and 72.22% of patients in the HD-MTX monotherapy group (p = 0.7221), while the overall response rate was 72% vs. 77.78% (p = 0.6063). Median PFS in HD-MTX/IDA group and HD-MTX monotherapy group were 15.6 months and 18.5 months, respectively (p = 0.6374). Median OS was not reached in both groups. There were no significant differences in adverse effects between the two groups.
Conclusions
The combination of IDA with HD-MTX showed no obvious therapeutic advantage over HD-MTX monotherapy in newly diagnosed patients with PCNSL. HD-MTX dose of 8 g/m2 monotherapy can still provide better therapeutic benefits in patients with acceptable adverse effects. Future studies could explore HD-MTX in combination with other chemotherapeutic agents in the first-line treatment of PCNSL.



Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Schaff LR, Grommes C (2021) Primary central nervous system lymphoma. Blood. https://doi.org/10.1182/blood.2020008377
Mendez JS, Ostrom QT, Gittleman H et al (2018) The elderly left behind-changes in survival trends of primary central nervous system lymphoma over the past 4 decades. Neuro Oncol 20:687–694
Chukwueke U, Grommes C, Nayak L (2022) Primary central nervous system lymphomas. Hematol Oncol Clin North Am 36:147–159
Alcantara M, Fuentealba J, Soussain C (2021) Emerging landscape of immunotherapy for primary central nervous system lymphoma. Cancers (Basel) 13:5061
Network. NCC. NCCN clinical practice guidelines in oncology: central nervous system cancers (Version1.2021). Available at https://www.nccn.org/professionals/physician_gls/pdf/cns.pdf. Accessed on 4 Jun 2021
Sieg N, Naendrup JH, Gödel P et al (2021) Treatment patterns and disease course of previously untreated primary central nervous system lymphoma: feasibility of MTX-based regimens in clinical routine. Eur J Haematol 107:202–210
van der Meulen M, Dirven L, Habets EJJ et al (2018) Cognitive functioning and health-related quality of life in patients with newly diagnosed primary CNS lymphoma: a systematic review. Lancet Oncol 19:e407–e418
Correa DD, Braun E, Kryza-Lacombe M et al (2019) Longitudinal cognitive assessment in patients with primary CNS lymphoma treated with induction chemotherapy followed by reduced-dose whole-brain radiotherapy or autologous stem cell transplantation. J Neurooncol 144:553–562
Yoon WS, Park JS, Kim YI et al (2021) High-dose methotrexate monotherapy for newly diagnosed primary central nervous system lymphoma: 15-year multicenter experience. Asia Pac J Clin Oncol 17:123–130
Liu Y, Yao Q, Zhang F (2021) Diagnosis, prognosis and treatment of primary central nervous system lymphoma in the elderly population (Review). Int J Oncol 58:371–387
Holdhoff M, Mrugala MM, Grommes C et al (2020) Challenges in the treatment of newly diagnosed and recurrent primary central nervous system lymphoma. J Natl Compr Canc Netw 18:1571–1578
Chihara D, Dunleavy K (2021) Primary central nervous system lymphoma: evolving biologic insights and recent therapeutic advances. Clin Lymphoma Myeloma Leuk 21:73–79
Ferreri AJ, Ciceri F, Brandes AA et al (2014) MATILDE chemotherapy regimen for primary CNS lymphoma: results at a median follow-up of 12 years. Neurology 82:1370–1373
Qian L, Zhou C, Shen J et al (2016) Treatment of newly diagnosed B-cell origin primary CNS lymphoma with systemic R-IDARAM chemotherapy and intrathecal immunochemotherapy. Oncotarget 7:25783–25790
Olivier G, Clavert A, Lacotte-Thierry L et al (2014) A phase 1 dose escalation study of idarubicin combined with methotrexate, vindesine, and prednisolone for untreated elderly patients with primary central nervous system lymphoma. The GOELAMS LCP 99 trial. Am J Hematol 89:1024–1029
Zhao D, Qian L, Shen J et al (2014) Combined treatment of rituximab, idarubicin, dexamethasone, cytarabine, methotrexate with radiotherapy for primary central nervous system lymphoma. J Cell Mol Med 18:1081–1086
Crivellari D, Lombardi D, Spazzapan S et al (2004) New oral drugs in older patients: a review of idarubicin in elderly patients. Crit Rev Oncol Hematol 49:153–163
Reid JM, Pendergrass TW, Krailo MD et al (1990) Plasma pharmacokinetics and cerebrospinal fluid concentrations of idarubicin and idarubicinol in pediatric leukemia patients: a Childrens Cancer Study Group report. Cancer Res 50:6525–6528
Fan N, Zhang L, Xu X et al (2017) Methotrexate plus idarubicin improves outcome of patients with primary central nervous system lymphoma. Oncotarget 8(32):53701–53713
Ferreri AJM, Blay J-Y, Reni M et al (2003) Prognostic scoring system for primary CNS lymphomas: the International Extranodal Lymphoma Study Group experience. J Clin Oncol 21:266–272
Thiel E, Korfel A, Martus P et al (2010) High-dose methotrexate with or without whole brain radiotherapy for primary CNS lymphoma (G-PCNSL-SG-1): a phase 3, randomised, non-inferiority trial. Lancet Oncol 11:1036–1047
Bromberg JEC, Issa S, Bakunina K et al (2019) Rituximab in patients with primary CNS lymphoma (HOVON 105/ALLG NHL 24): a randomised, open-label, phase 3 intergroup study. Lancet Oncol 20:216–228
Ferreri AJM, Cwynarski K, Pulczynski E et al (2016) Chemoimmunotherapy with methotrexate, cytarabine, thiotepa, and rituximab (MATRix regimen) in patients with primary CNS lymphoma: results of the first randomisation of the International Extranodal Lymphoma Study Group-32 (IELSG32) phase 2 trial. Lancet Haematol 3:e217–e227
Ferreri AJ, Reni M, Foppoli M et al (2009) High-dose cytarabine plus high-dose methotrexate versus high-dose methotrexate alone in patients with primary CNS lymphoma: a randomised phase 2 trial. Lancet 374:1512–1520
Omuro A, Chinot O, Taillandier L et al (2015) Methotrexate and temozolomide versus methotrexate, procarbazine, vincristine, and cytarabine for primary CNS lymphoma in an elderly population: an intergroup ANOCEF-GOELAMS randomised phase 2 trial. Lancet Haematol 2:e251–e259
Brezina T, von Dewitz H, Schroeder T et al (2022) First-line high-dose therapy and autologous blood stem cell transplantation in patients with primary central nervous system non-Hodgkin lymphomas-a single-centre experience in 61 patients. Ann Hematol 101:607–616
Gritsch D, Mrugala MM, Marks LA et al (2021) Is autologous stem cell transplantation a safe and effective alternative to whole brain radiation as consolidation therapy in patients with primary central nervous system lymphoma?: A critically appraised topic. Neurologist 26:137–142
Choi YS (2020) Recent advances in the management of primary central nervous system lymphoma. Blood Res 55:S58-s62
Seidel S, Margold M, Kowalski T et al (2021) Patients with primary central nervous system lymphoma not eligible for clinical trials: prognostic factors, treatment and outcome. Cancers (Basel) 13:2934
Fox CP, Phillips EH, Smith J et al (2019) Guidelines for the diagnosis and management of primary central nervous system diffuse large B-cell lymphoma. Br J Haematol 184:348–363
Yuan Y, Ding T, Wang S et al (2021) Current and emerging therapies for primary central nervous system lymphoma. Biomark Res 9:32
Li Q, Ma J, Ma Y et al (2021) Improvement of outcomes of an escalated high-dose methotrexate-based regimen for patients with newly diagnosed primary central nervous system lymphoma: a real-world cohort study. Cancer Manag Res 13:6115–6122
Acknowledgements
Not applicable.
Funding
Shanghai Shenkang Clinical Innovation Project (Project No. SHDC12020112). Beijing Medical and Health Foundation (Project No. YWJKJJHKB175B). Clinical Research Plan of SHDC (SHDC 2020CR6005-002).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study’s conception and design. Data collection and data analysis were performed by QL and YM. The first draft of the manuscript was written by QL and BC, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest regarding the publication of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Qing Li and Yan Ma are contributed equally to this work.
Rights and permissions
About this article
Cite this article
Li, Q., Ma, Y., Lin, Z. et al. A prospective cohort study of methotrexate plus idarubicin in newly diagnosed primary CNS lymphoma. J Neurooncol 163, 39–46 (2023). https://doi.org/10.1007/s11060-022-04062-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-022-04062-z