Abstract
Background
Neoadjuvant chemotherapy (NAC) or chemoradiation (NAC+XRT) is incorporated into the treatment of localized pancreatic adenocarcinoma (PDAC), often with the goal of downstaging before resection. However, the effect of downstaging on overall survival, particularly the differential effects of NAC and NAC+XRT, remains undefined. This study examined the impact of downstaging from NAC and NAC+XRT on overall survival.
Methods
The National Cancer Data Base (NCDB) was queried from 2006 to 2015 for patients with non-metastatic PDAC who received NAC or NAC+XRT. Rates of overall and nodal downstaging, and pathologic complete response (pCR) were assessed. Predictors of downstaging were evaluated using multivariable logistic regression. Overall survival (OS) was assessed with Kaplan–Meier and Cox proportional hazards modeling.
Results
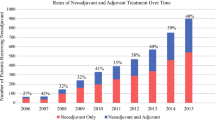
The study enrolled 2475 patients (975 NAC and 1500 NAC+XRT patients). Compared with NAC, NAC+XRT was associated with higher rates of overall downstaging (38.3 % vs 23.6 %; p ≤ 0.001), nodal downstaging (16.0 % vs 7.8 %; p ≤ 0.001), and pCR (1.7 % vs 0.7 %; p = 0.041). Receipt of NAC+XRT was independently predictive of overall (odds ratio [OR] 2.28; p < 0.001) and nodal (OR 3.09; p < 0.001) downstaging. Downstaging by either method was associated with improved 5-year OS (30.5 vs 25.2 months; p ≤ 0.001). Downstaging with NAC was associated with an 8-month increase in median OS (33.7 vs 25.6 months; p = 0.005), and downstaging by NAC+XRT was associated with a 5-month increase in median OS (30.0 vs 25.0 months; p = 0.008). Cox regression showed an association of overall downstaging with an 18 % reduction in the risk of death (hazard ratio [HR] 0.82; 95 % confidence interval, 0.71–0.95; p = 0.01)
Conclusion
Downstaging after neoadjuvant therapies improves survival. The addition of radiation therapy may increase the rate of downstaging without affecting overall oncologic outcomes.



Similar content being viewed by others
References
Ryan DP, Hong TS, Bardeesy N. Pancreatic adenocarcinoma. N Engl J Med. 2014;371:1039–49. https://doi.org/10.1056/NEJMra1404198.
Yamamoto T, Yagi S, Kinoshita H, et al. Long-term survival after resection of pancreatic cancer: a single-center retrospective analysis. World J Gastroenterol. 2015;21:262–8. https://doi.org/10.3748/wjg.v21.i1.262.
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020 (in English). CA Cancer J Clin. 2020;70:7–30. https://doi.org/10.3322/caac.21590.
National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology: Pancreatic Adenocarcinoma. Retrieved 25 February 2021 at https://www.nccn.org/professionals/physician_gls/pdf/pancreatic.pdf.
Balaban EP, Mangu PB, Khorana AA, et al. Locally advanced, unresectable pancreatic cancer: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2016;34:2654–68. https://doi.org/10.1200/jco.2016.67.5561.
White RR, Hurwitz HI, Morse MA, et al. Neoadjuvant chemoradiation for localized adenocarcinoma of the pancreas. Ann Surg Oncol. 2001;8:758–65. https://doi.org/10.1007/s10434-001-0758-1.
Palmer DH, Stocken DD, Hewitt H, et al. A randomized phase 2 trial of neoadjuvant chemotherapy in resectable pancreatic cancer: gemcitabine alone versus gemcitabine combined with cisplatin. Ann Surg Oncol. 2007;14:2088–96. https://doi.org/10.1245/s10434-007-9384-x.
Evans DB, Varadhachary GR, Crane CH, et al. Preoperative gemcitabine-based chemoradiation for patients with resectable adenocarcinoma of the pancreatic head. J Clin Oncol. 2008;26:3496–502. https://doi.org/10.1200/JCO.2007.15.8634.
Katz MH, Shi Q, Ahmad SA, et al. Preoperative modified FOLFIRINOX treatment followed by capecitabine-based chemoradiation for borderline resectable pancreatic cancer: Alliance for Clinical Trials in Oncology Trial A021101. JAMA Surg. 2016;151:e161137. https://doi.org/10.1001/jamasurg.2016.1137.
Gillen S, Schuster T, Meyer Zum Buschenfelde C, Friess H, Kleeff J. Preoperative/neoadjuvant therapy in pancreatic cancer: a systematic review and meta-analysis of response and resection percentages. PLoS Med. 2010;7:e1000267. https://doi.org/10.1371/journal.pmed.1000267.
Schwarz L, Vernerey D, Bachet JB, et al. Resectable pancreatic adenocarcinoma neoadjuvant FOLF(IRIN)OX-based chemotherapy: a multicenter, non-comparative, randomized, phase II trial (PANACHE01-PRODIGE48 study). BMC Cancer. 2018;18:762. https://doi.org/10.1186/s12885-018-4663-4.
Zhan HX, Xu JW, Wu D, et al. Neoadjuvant therapy in pancreatic cancer: a systematic review and meta-analysis of prospective studies. Cancer Med. 2017;6:1201–19. https://doi.org/10.1002/cam4.1071.
Hu Q, Wang D, Chen Y, Li X, Cao P, Cao D. Network meta-analysis comparing neoadjuvant chemoradiation, neoadjuvant chemotherapy, and upfront surgery in patients with resectable, borderline resectable, and locally advanced pancreatic ductal adenocarcinoma. Radiat Oncol. 2019;14:120. https://doi.org/10.1186/s13014-019-1330-0.
Tzeng CW, Balachandran A, Ahmad M, et al. Serum carbohydrate antigen 19–9 represents a marker of response to neoadjuvant therapy in patients with borderline resectable pancreatic cancer. HPB Oxford. 2014;16:430–8. https://doi.org/10.1111/hpb.12154.
Cacciato Insilla A, Vivaldi C, Giordano M, et al. Tumor regression grading assessment in locally advanced pancreatic cancer after neoadjuvant FOLFIRINOX: interobserver agreement and prognostic implications. Front Oncol. 2020;10:64. https://doi.org/10.3389/fonc.2020.00064.
Chatterjee D, Katz MH, Rashid A, et al. Histologic grading of the extent of residual carcinoma following neoadjuvant chemoradiation in pancreatic ductal adenocarcinoma: a predictor for patient outcome. Cancer. 2012;118:3182–90. https://doi.org/10.1002/cncr.26651.
Sugimoto M, Takahashi N, Farnell MB, et al. Survival benefit of neoadjuvant therapy in patients with non-metastatic pancreatic ductal adenocarcinoma: a propensity-matching and intention-to-treat analysis. J Surg Oncol. 2019;120:976–84. https://doi.org/10.1002/jso.25681.
Michelakos T, Pergolini I, Castillo CF, et al. Predictors of resectability and survival in patients with borderline and locally advanced pancreatic cancer who underwent neoadjuvant treatment with FOLFIRINOX. Ann Surg. 2019;269:733–40. https://doi.org/10.1097/SLA.0000000000002600.
Schorn S, Demir IE, Reyes CM, et al. The impact of neoadjuvant therapy on the histopathological features of pancreatic ductal adenocarcinoma: a systematic review and meta-analysis. Cancer Treat Rev. 2017;55:96–106. https://doi.org/10.1016/j.ctrv.2017.03.003.
Hammel P, Huguet F, van Laethem JL, et al. Effect of chemoradiotherapy vs chemotherapy on survival in patients with locally advanced pancreatic cancer controlled after 4 months of gemcitabine with or without erlotinib: the LAP07 Randomized Clinical Trial. JAMA. 2016;315:1844–53. https://doi.org/10.1001/jama.2016.4324.
Katz MH, Kim MP, Tzeng CW, Lee JE. Preoperative chemoradiation for borderline resectable pancreatic cancer: the new standard? Ann Surg. 2018;268:223–4. https://doi.org/10.1097/SLA.0000000000002783.
Jang JY, Han Y, Lee H, et al. Oncological benefits of neoadjuvant chemoradiation with gemcitabine versus upfront surgery in patients with borderline resectable pancreatic cancer: a prospective, randomized, open-label, multicenter phase 2/3 trial. Ann Surg. 2018;268:215–22. https://doi.org/10.1097/SLA.0000000000002705.
Eijck CHJV, Versteijne E, Suker M, et al. Preoperative chemoradiotherapy to improve overall survival in pancreatic cancer: long-term results of the multicenter randomized phase III PREOPANC trial. J Clin Oncol. 2021;39(15 Suppl):4016. https://doi.org/10.1200/JCO.2021.39.15_suppl.4016.
Katz MHG, Shi Q, Meyers JP, et al. Alliance A021501: preoperative mFOLFIRINOX or mFOLFIRINOX plus hypofractionated radiation therapy (RT) for borderline resectable (BR) adenocarcinoma of the pancreas. J Clin Oncol. 2021;39(3 Suppl):377. https://doi.org/10.1200/JCO.2021.39.3_suppl.377.
Loehrer PJ Sr, Feng Y, Cardenes H, et al. Gemcitabine alone versus gemcitabine plus radiotherapy in patients with locally advanced pancreatic cancer: an Eastern Cooperative Oncology Group trial. J Clin Oncol. 2011;29:4105–12. https://doi.org/10.1200/JCO.2011.34.8904.
Chauffert B, Mornex F, Bonnetain F, et al. Phase III trial comparing intensive induction chemoradiotherapy (60 Gy, infusional 5-FU and intermittent cisplatin) followed by maintenance gemcitabine with gemcitabine alone for locally advanced unresectable pancreatic cancer: definitive results of the 2000–01 FFCD/SFRO study. Ann Oncol. 2008;19:1592–9. https://doi.org/10.1093/annonc/mdn281.
Bilimoria KY, Stewart AK, Winchester DP, Ko CY. The National Cancer Data Base: a powerful initiative to improve cancer care in the United States. Ann Surg Oncol. 2008;15:683–90. https://doi.org/10.1245/s10434-007-9747-3.
Hill C, Rosati LM, Hu C, et al. Long-term outcomes with neoadjuvant chemotherapy with or without stereotactic body radiation therapy in patients with borderline resectable and locally advanced pancreatic adenocarcinoma. J Clin Oncol. 2021;39(3 Suppl):443. https://doi.org/10.1200/JCO.2021.39.3_suppl.443.
Katz MHG, Ou FS, Herman JM, et al. Alliance for clinical trials in oncology (ALLIANCE) trial A021501: preoperative extended chemotherapy vs chemotherapy plus hypofractionated radiation therapy for borderline resectable adenocarcinoma of the head of the pancreas. BMC Cancer. 2017;17:505. https://doi.org/10.1186/s12885-017-3441-z.
Oba A, Ho F, Bao QR, Al-Musawi MH, Schulick RD, Del Chiaro M. Neoadjuvant treatment in pancreatic cancer (in English). Front Oncol. 2020;10:245. https://doi.org/10.3389/fonc.2020.00245.
Poruk KE, Gay DZ, Brown K, et al. The clinical utility of CA 19–9 in pancreatic adenocarcinoma: diagnostic and prognostic updates (in English). Curr Mol Med. 2013;13:340–51. https://doi.org/10.2174/1566524011313030003.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
There are no conflicts of interest.
NCDB Statement
The NCDB is a joint project of the Commission on Cancer of the American College of Surgeons and the American Cancer Society. The data used in the study are derived from a de-identified NCDB file. The American College of Surgeons and the Commission on Cancer have not verified and are not responsible for the analytic or statistical methodology used or the conclusions drawn from these data by the investigator.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
O’Shea, A.E., Bohan, P.M.K., Carpenter, E.L. et al. Downstaging of Pancreatic Adenocarcinoma With Either Neoadjuvant Chemotherapy or Chemoradiotherapy Improves Survival. Ann Surg Oncol 29, 6015–6028 (2022). https://doi.org/10.1245/s10434-022-11800-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-022-11800-0