Abstract
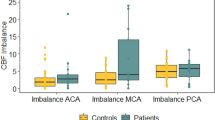
Moyamoya disease is a major arteriopathy characterised by progressive steno-occlusion of the arteries of the circle of Willis. Studies in adults with moyamoya suggest an association between abnormal fronto-parietal and white matter regional haemodynamics and cognitive impairments, even in the absence of focal infarction. However, these associations have not been investigated in children with moyamoya. We examined the relationship between regional haemodynamics and ratings of intellectual ability and executive function, using hypercapnic challenge blood oxygen level–dependent magnetic resonance imaging of cerebrovascular reactivity in a consecutive cohort of children with confirmed moyamoya. Thirty children were included in the final analysis (mean age: 12.55 ± 3.03 years, 17 females, 15 idiopathic moyamoya and 15 syndromic moyamoya). Frontal haemodynamics were abnormal in all regardless of stroke history and comorbidity, but occipital lobe haemodynamics were also abnormal in children with syndromic moyamoya. Executive function deficits were noted in both idiopathic and syndromic moyamoya, whereas intellectual ability was impaired in syndromic moyamoya, even in the absence of stroke. Analysis of the relative effect of regional abnormal haemodynamics on cognitive outcomes demonstrated that executive dysfunction was predominantly explained by right parietal and white matter haemodynamics independent of stroke and comorbidity, while posterior circulation haemodynamics predicted intellectual ability. These results suggest that parietal and posterior haemodynamics play a compensatory role in overcoming frontal vulnerability and cognitive impairment.


Similar content being viewed by others
Availability of Data and Material
The datasets of the current study are not publicly available due to privacy and ethical concerns but are available from the corresponding author on reasonable request.
Code Availability
Not applicable.
References
Ikezaki K, Matsushima T, Kuwabara Y, Suzuki S, Nomura T, Fukui M. Cerebral circulation and oxygen metabolism in childhood moyamoya disease: a perioperative positron emission tomography study. J Neurosurg. 1994;81:843–50.
Grant DA, Franzini C, Wild J, Eede KJ, Walker AM. Autoregulation of the cerebral circulation during sleep in newborn lambs. J Physiol. 2005;564:923–30 (John Wiley & Sons, Ltd).
Derdeyn CP, Shaibani A, Moran CJ, Cross DT, Grubb RL, Powers WJ. Lack of correlation between pattern of collateralization and misery perfusion in patients with carotid occlusion. Stroke Lippincott Williams and Wilkins. 1999;30:1025–32.
Derdeyn C, Videen T, Yundt K, Fritsch S, Carpenter D, Grubb R, et al. Variability of cerebral blood volume and oxygen extraction: stages of cerebral haemodynamic impairment revisited. Brain. 2002;125:595–607.
Baron JC, Bousser MG, Rey A, Guillard A, Comar D, Castaigne P. Reversal of focal “misery-perfusion syndrome” by extra-intracranial arterial bypass in hemodynamic cerebral ischemia. A case study with 15O positron emission tomography. Stroke. 1981;12:454–9.
Ohtaki M, Uede T, Morimoto S, Nonaka T, Tanabe S, Hashi K. Intellectual functions and regional cerebral haemodynamics after extensive omental transplantation spread over both frontal lobes in childhood moyamoya disease. Acta Neurochir (Wien). 1998;140:1043–53 (Springer).
Williams TS, Westmacott R, Dlamini N, Granite L, Dirks P, Askalan R, et al. Intellectual ability and executive function in pediatric moyamoya vasculopathy. Dev Med Child Neurol. 2012;54:30–7.
Isono M, Ishii K, Kamida T, Inoue R, Fujiki M, Kobayashi H. Long-term outcomes of pediatric moyamoya disease treated by encephalo-duro-arterio-synangiosis. Pediatr Neurosurg. 2002;36:14–21.
Hogan AM, Kirkham FJ, Isaacs EB, Wade AM, Vargha-Khadem F. Intellectual decline in children with moyamoya and sickle cell anaemia. Dev Med Child Neurol. 2005;47:824–9.
Ishii R, Takeuchi S, Tanaka R (1984) Intelligence in children with moyamoya disease: evaluation after surgical treatments with special reference to changes in cerebral blood flow. Stroke
Imaizumi T, Hayashi K, Saito K, Osawa M, Fukuyama Y. Long-term outcomes of pediatric moyamoya disease monitored to adulthood. Pediatr Neurol. 1998;18:321–5.
Imaizumi C, Imaizumi T, Osawa M, Fukuyama Y, Takeshita M. Serial intelligence test scores in pediatric moyamoya disease. Neuropediatrics. 1999;30:294–9.
Lee JY, Phi JH, Wang K-C, Cho B-K, Shin M-S, Kim S-K. Neurocognitive profiles of children with moyamoya disease before and after surgical intervention. Cerebrovasc Dis. 2011;31:230–7.
Matsushima Y, Aoyagi M, Nariai T, … YT-C neurology and, 1997 U. Long-term intelligence outcome of post-encephalo-duro-arterio-synangiosis childhood moyamoya patients. Clin Neurol Neurosurg. 1997;99:S147–50.
Matsushima Y, Aoyagi M, Masaoka H, Suzuki R, Ohno K. Mental outcome following encephaloduroarteriosynangiosis in children with moyamoya disease with the onset earlier than 5 years of age. Childs Nerv Syst. 1990;6:440–3.
Bowen M, Marks MP, Steinberg GK. Neuropsychological recovery from childhood moyamoya disease. Brain Dev Elsevier. 1998;20:119–23.
Karzmark P, Zeifert PD, Tan S, Dorfman LJ, Bell-Stephens TE, Steinberg GK. Effect of moyamoya disease on neuropsychological functioning in adults. Neurosurgery. 2008;62:1048–51.
Ibrahim AFA, Montojo CA, Haut KM, Karlsgodt KH, Hansen L, Congdon E, et al. Spatial working memory in neurofibromatosis 1: altered neural activity and functional connectivity. NeuroImage Clin. 2017;15:801–11 (Elsevier Inc.).
Payne JM, Hyman SL, Shores EA, North KN. Assessment of executive function and attention in children with neurofibromatosis type 1: relationships between cognitive measures and real-world behavior. Child Neuropsychol. 2011;17:313–29.
Hijmans CT, Fijnvandraat K, Grootenhuis MA, van Geloven N, Heijboer H, Peters M, et al. Neurocognitive deficits in children with sickle cell disease: a comprehensive profile. Pediatr Blood Cancer. 2011;56:783–8 (John Wiley & Sons, Ltd).
Karzmark P, Zeifert PD, Bell-Stephens TE, Steinberg GK, Dorfman LJ. Neurocognitive impairment in adults with moyamoya disease without stroke. Neurosurgery Oxford University Press (OUP). 2012;70:634–8.
Quon JL, Kim LH, MacEachern SJ, Maleki M, Steinberg GK, Madhugiri V, et al. Early diffusion magnetic resonance imaging changes in normal-appearing brain in pediatric moyamoya disease. Neurosurgery Oxford University Press. 2020;86:530–7.
Dlamini N, Shah-Basak P, Leung J, Kirkham F, Shroff M, Kassner A, et al. Breath-hold blood oxygen level-dependent MRI: a tool for the assessment of cerebrovascular reserve in children with moyamoya disease. Am J Neuroradiol. 2018;39:1717–23.
Spano VR, Mandell DM, Poublanc J, Sam K, Battisti-Charbonney A, Pucci O, et al. CO2 blood oxygen level-dependent MR mapping of cerebrovascular reserve in a clinical population: safety, tolerability, and technical feasibility. Radiology. 2013;266:592–8.
Pillai JJ, Mikulis DJ. Cerebrovascular reactivity mapping: an evolving standard for clinical functional imaging. Am. J. Neuroradiol. American Society of Neuroradiology; 2015. p. 7–13.
Gupta A, Chazen JL, Hartman M, Delgado D, Anumula N, Shao H, et al. Cerebrovascular reserve and stroke risk in patients with carotid stenosis or occlusion: a systematic review and meta-analysis. Stroke. 2012. p. 2884–91.
Jackman K, Iadecola C. Neurovascular regulation in the ischemic brain. Antioxidants Redox Signal. Mary Ann Liebert Inc.; 2015. p. 149–60.
Yonas H, Pindzola RR, Meltzer CC, Sasser H. Qualitative versus quantitative assessment of cerebrovascular reserves. Neurosurgery Lippincott Williams and Wilkins. 1998;42:1005–12.
Ogasawara K, Okuguchi T, Sasoh M, Kobayashi M, Yukawa H, Terasaki K, et al. Qualitative versus quantitative assessment of cerebrovascular reactivity to acetazolamide using iodine-123-N-isopropyl-p-iodoamphetamine SPECT in patients with unilateral major cerebral artery occlusive disease. AJNR Am J Neuroradiol. 2003;24:1090–5.
Dlamini N, Slim M, Kirkham F, Shroff M, Dirks P, Moharir M, et al. Predicting Ischemic Risk Using Blood Oxygen Level-Dependent MRI in Children with Moyamoya. Am J Neuroradiol. 2020;41:160–6.
Heyn C, Poublanc J, Crawley A, Mandell D, Han JS, Tymianski M, et al. Quantification of cerebrovascular reactivity by blood oxygen level-dependent mr imaging and correlation with conventional angiography in patients with moyamoya disease. Am J Neuroradiol. 2010;
Han JS, Mikulis DJ, Mardimae A, Kassner A, Poublanc J, Crawley AP, et al. Measurement of cerebrovascular reactivity in pediatric patients with cerebral vasculopathy using blood oxygen level-dependent MRI. Stroke. 2011;42:1261–9.
Dlamini N, Yau I, Westmacott R, Shroff M, Armstrong D, Logan W, et al. Cerebrovascular reactivity and intellectual outcome in childhood stroke with transient cerebral arteriopathy. Pediatr Neurol Elsevier. 2017;69:71–8.
Hashimoto N, Tominaga T, Miyamoto S, Nagata I, Houkin K, Suzuki N, et al. Guidelines for diagnosis and treatment of moyamoya disease (spontaneous occlusion of the circle of Willis). Neurol Med Chir (Tokyo). Japan Neurosurgical Society; 2012;52:245–66.
Rafay MF, Armstrong D, Dirks P, MacGregor DL, DeVeber G. Patterns of cerebral ischemia in children with moyamoya. Pediatr Neurol Elsevier. 2015;52:65–72.
Suzuki J, Takaku A. Cerebrovascular moyamoya disease: Disease showing abnormal net-like vessels in base of brain. Arch Neurol American Medical Association. 1969;20:288–99.
Wechsler D. Wechsler intelligence scale for children. 3rd ed. San Antonio, TX: The Psychological Corporation; 1991.
Wechsler D. Wechsler intelligence scale for children (WISC). 4th edn. San Antonio, TX: Harcourt Assessment; 2004.
Wechsler D. The Wechsler adult intelligence scale. 3rd ed. San Antonio, TX: The Psychological Corporation; 1997.
Gioia G, Isquith P, Guy S, Kenworthy L. BRIEF: behavior rating inventory of executive function professional manual. Lutz, FL: Psychological Assessment Resources, Inc; 2000.
Mugikura S, Takahashi S, Higano S, Shirane R, Sakurai Y, Yamada S. Predominant involvement of ipsilateral anterior and posterior circulations in moyamoya disease. Stroke. 2002;33:1497–500.
Tomasi D, Volkow ND. Resting functional connectivity of language networks: characterization and reproducibility. Mol Psychiatry Nature Publishing Group. 2012;17:841–54.
Kang CG, Chun MH, Kang JA, Do KH, Choi SJ. Neurocognitive dysfunction according to hypoperfusion territory in patients with moyamoya disease. Ann Rehabil Med. Korean Academy of Rehabilitation Medicine; 2017;41:1–8.
Calviere L, Catalaa I, Marlats F, Viguier A, Bonneville F, Cognard C, et al. Correlation between cognitive impairment and cerebral hemodynamic disturbances on perfusion magnetic resonance imaging in European adults with moyamoya disease. J Neurosurg. 2010;113:753–9.
Su J Bin, Xi S Da, Zhou SY, Zhang X, Jiang SH, Xu B, et al. Microstructural damage pattern of vascular cognitive impairment: a comparison between moyamoya disease and cerebrovascular atherosclerotic disease. Neural Regen Res. Wolters Kluwer Medknow Publications; 2019;14:858–67.
Fiske A, Holmboe K. Neural substrates of early executive function development. Dev. Rev. Mosby Inc.; 2019. p. 42–62.
Kalpakidou AK, Allin MPG, Walshe M, Giampietro V, McGuire PK, Rifkin L, et al. Functional neuroanatomy of executive function after neonatal brain injury in adults who were born very preterm. PLoS One. Public Library of Science; 2014;9.
Woodward LJ, Clark CAC, Pritchard VE, Anderson PJ, Inder TE. Neonatal white matter abnormalities predict global executive function impairment in children born very preterm. Dev Neuropsychol Taylor & Francis Group. 2011;36:22–41.
Wang Q, Cheung C, Deng W, Li M, Huang C, Ma X, et al. Fronto-parietal white matter microstructural deficits are linked to performance IQ in a first-episode schizophrenia Han Chinese sample. Psychol Med Psychol Med. 2013;43:2047–56.
Ohtani T, Nestor PG, Bouix S, Newell D, Melonakos ED, McCarley RW, et al. Exploring the neural substrates of attentional control and human intelligence: diffusion tensor imaging of prefrontal white matter tractography in healthy cognition. Neuroscience Elsevier Ltd. 2017;341:52–60.
Burgess PW, Stuss DT. Fifty years of prefrontal cortex research: impact on assessment. J Int Neuropsychol Soc. Cambridge University Press; 2017;23:755–67.
Ohtani T, Nestor PG, Bouix S, Saito Y, Hosokawa T, Kubicki M. Medial frontal white and gray matter contributions to general intelligence. PLoS One. Public Library of Science; 2014;9.
Goh S, Bansal R, Xu D, Hao X, Liu J, Peterson BS. Neuroanatomical correlates of intellectual ability across the life span. Dev Cogn Neurosci Elsevier. 2011;1:305–12.
Sasagawa A, Mikami T, Hirano T, Akiyama Y, Mikuni N. Characteristics of cerebral hemodynamics assessed by CT perfusion in moyamoya disease. J Clin Neurosci Churchill Livingstone. 2018;47:183–9.
Robinson H, Calamia M, Gläscher J, Bruss J, Tranel D. Neuroanatomical correlates of executive functions: a neuropsychological approach using the EXAMINER battery. J Int Neuropsychol Soc. Cambridge University Press; 2014;20:52–63.
Hampshire A, Chamberlain SR, Monti MM, Duncan J, Owen AM. The role of the right inferior frontal gyrus: inhibition and attentional control. Neuroimage Academic Press. 2010;50:1313–9.
Spagna A, Kim TH, Wu T, Fan J. Right hemisphere superiority for executive control of attention. Cortex Masson SpA. 2020;122:263–76.
Luders E, Narr KL, Thompson PM, Toga AW. Neuroanatomical correlates of intelligence. Intelligence NIH Public Access. 2009;37:156–63.
Wechsler D. The measurement of adult intelligence. Meas. adult Intell. (3rd ed.). Williams & Wilkins Co; 2007.
Damasio A, Anderson S. The frontal lobes. In: Heilman K, Valenstein E, editors. Clin Neuropsychol. Oxford University Press; 2010. p. 404–46.
Ardila A, Pineda D, Rosselli M. Correlation between intelligence test scores and executive function measures. Arch Clin Neuropsychol. 2000;15:31–6.
Ehrler M, Latal B, Kretschmar O, von Rhein M, O’Gorman Tuura R. Altered frontal white matter microstructure is associated with working memory impairments in adolescents with congenital heart disease: a diffusion tensor imaging study. NeuroImage Clin. Elsevier Inc.; 2020;25.
Kazumata K, Tha KK, Narita H, Kusumi I, Shichinohe H, Ito M, et al. Chronic ischemia alters brain microstructural integrity and cognitive performance in adult moyamoya disease. Stroke Lippincott Williams and Wilkins. 2015;46:354–60.
Stotesbury H, Kirkham FJ, Kölbel M, Balfour P, Clayden JD, Sahota S, et al. White matter integrity and processing speed in sickle cell anemia. Neurology Neurology. 2018;90:E2042–50.
Krukow P, Jonak K, Karakuła-Juchnowicz H, Podkowiński A, Jonak K, Borys M, et al. Disturbed functional connectivity within the left prefrontal cortex and sensorimotor areas predicts impaired cognitive speed in patients with first-episode schizophrenia. Psychiatry Res - Neuroimaging. 2018;275:28–35 (Elsevier Ireland Ltd).
Gayda M, Gremeaux V, Bherer L, Juneau M, Drigny J, Dupuy O, et al. Cognitive function in patients with stable coronary heart disease: related cerebrovascular and cardiovascular responses. PLoS One. Public Library of Science; 2017;12.
Gläscher J, Tranel D, Paul LK, Rudrauf D, Rorden C, Hornaday A, et al. Lesion mapping of cognitive abilities linked to intelligence. Neuron Neuron. 2009;61:681–91.
Lee M, Zaharchuk G, Guzman R, Achrol A, Bell-Stephens T, Steinberg GK. Quantitative hemodynamic studies in moyamoya disease. Neurosurg Focus. Journal of Neurosurgery Publishing Group (JNSPG); 2009;26:E5.
Baltsavias G, Khan N, Valavanis A. The collateral circulation in pediatric moyamoya disease. Child’s Nerv Syst Springer Verlag. 2015;31:389–98.
Suzuki J, Takaku A. Cerebrovascular, “moyamoya” disease: disease showing abnormal net-like vessels in base of brain. Arch Neurol. 1969;20:288–99.
Wiesman AI, Wilson TW. The impact of age and sex on the oscillatory dynamics of visuospatial processing. Neuroimage. 2019;185:513–20 (Academic Press Inc.).
Billingsley RL, Jackson EF, Slopis JM, Swank PR, Mahankali S, Moore BD. Functional MRI of visual-spatial processing in neurofibromatosis, type I. Neuropsychologia Elsevier Ltd. 2004;42:395–404.
Clements-Stephens AM, Rimrodt SL, Gaur P, Cutting LE. Visuospatial processing in children with neurofibromatosis type 1. Neuropsychologia NIH Public Access. 2008;46:690–7.
Yeom KW, Lober RM, Barnes PD, Campen CJ. Reduced cerebral arterial spin-labeled perfusion in children with neurofibromatosis type 1. Am J Neuroradiol. American Society of Neuroradiology; 2013. p. 1823–8.
Hyman SL, Shores A, North KN. The nature and frequency of cognitive deficits in children with neurofibromatosis type 1. Neurology Lippincott Williams and Wilkins. 2005;65:1037–44.
Vogel AC, Gutmann DH, Morris SM. Neurodevelopmental disorders in children with neurofibromatosis type 1. Dev Med Child Neurol. 2017;59:1112–6 (Blackwell Publishing Ltd).
Moses WW. Fundamental limits of spatial resolution in PET. Nucl Instrum Methods Phys Res A. NIH Public Access; 2011;648 Supplement 1:S236.
Purkayastha S, Sorond F. Transcranial Doppler ultrasound: technique and application. Semin Neurol Thieme Medical Publishers. 2012;32:411–20.
Funding
We thank the Auxilium Foundation and Brain Canada for their support. Study funders were not involved in the study design, data analysis, or publication decisions. The findings are solely the responsibility of the authors and do not represent the Auxilium Foundation. Image processing and analysis was supported by the Stroke Imaging Laboratory for Children, The Hospital for Sick Children, Toronto.
Author information
Authors and Affiliations
Contributions
The study concept and design were developed by Nomazulu Dlamini, William Logan, Gabrielle deVeber and Fenella J. Kirkham. The imaging and clinical data were collected by Nomazulu Dlamini, Prakash Muthusami, Mahendranath Moharir, Elizabeth Pulcine, Manohar Shroff, Peter Dirks, Daune MacGregor, Gabrielle deVeber, Matsanga Leyila Kaseka, Amanda Robertson and Ishvinder Bhathal. The clinical information was coded and analysed by Nomazulu Dlamini and Matsanga Leyila Kaseka. The neuropsychological data were collected by Robyn Westmacott and Tricia Williams. Eun Jung Choi analysed the data and wrote the paper. Mahmoud Slim reviewed the statistical methods. Nomazulu Dlamini, Fenella Kirkham, Robyn Westmacott, Prakash Muthusami, Mahendranath Moharir, Tricia Williams, Mahmoud Slim, Matsanga Leyila Kaseka, Elizabeth Pulcine, Andrea Kassner and William Logan reviewed and edited the paper.
Corresponding author
Ethics declarations
Ethics Approval
All procedures were approved by the Institutional Research Ethics Board at The Hospital for Sick Children.
Consent to Participate
Informed consent was obtained. All data were collected retrospectively, and patient identifiers were removed prior to analysis.
Consent to Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Choi, E.J., Westmacott, R., Kirkham, F.J. et al. Fronto-Parietal and White Matter Haemodynamics Predict Cognitive Outcome in Children with Moyamoya Independent of Stroke. Transl. Stroke Res. 13, 757–773 (2022). https://doi.org/10.1007/s12975-022-01003-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-022-01003-w