Abstract
Background
The foremost cause of death of breast cancer (BC) patients is metastasis, and the first site to which BC predominantly metastasizes is the axillary lymph node (ALN). Thus, ALN status is a key prognostic indicator at diagnosis. The immune system has an essential role in cancer progression and dissemination, so its evaluation in ALNs could have significant applications. In the present study we aimed to investigate the association of clinical-pathological and immune variables in the primary tumour and non-metastatic ALNs (ALNs–) of a cohort of luminal A and triple-negative BC (TNBC) patients with cancer-specific survival (CSS) and time to progression (TTP).
Methods
We analysed the differences in the variables between patients with different outcomes, created univariate and multivariate Cox regression models, validated them by bootstrapping and multiple imputation of missing data techniques, and used Kaplan–Meier survival curves for a 10-years follow-up.
Results
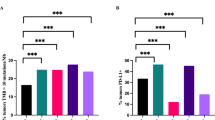
We found some clinical-pathological variables at diagnosis (tumour diameter, TNBC molecular profile and presence of ALN metastasis), and the levels of several immune markers in the two studied sites, to be associated with worse CSS and TTP. Nevertheless, only CD68 and CD83 in ALNs– were confirmed as independent prognostic factors for TTP.
Conclusions
The study identified the importance of macrophage and dendritic cell markers as prognostic factors of relapse for BC. We highlight the importance of studying the immune response in ALNs–, which could be relevant to the prediction of BC patients’ outcome.


Similar content being viewed by others
Availability of data and material
Datasets generated and analysed during this study are available from the corresponding authors upon reasonable request.
Abbreviations
- ALN:
-
Axillary lymph node
- ALN– :
-
Non-metastatic axillary lymph node
- ALN+ :
-
Metastatic axillary lymph node
- AUC:
-
Area under the curve
- BC:
-
Breast cancer
- CI:
-
Confidence interval
- CSS:
-
Cancer-specific survival
- DAB:
-
Diaminobenzidine
- DC:
-
Dendritic cell
- ER:
-
Oestrogen receptor
- HER2:
-
Human epidermal growth factor receptor 2
- HR:
-
Hazard ratio
- HTVC:
-
Hospital de Tortosa Verge de la Cinta
- Ki67:
-
Proliferation index
- NK:
-
Natural killer
- PR:
-
Progesterone receptor
- ROC:
-
Receiver-operating characteristic
- STROBE:
-
Strengthening the Reporting of Observational Studies in Epidemiology
- TMA:
-
Tissue microarray
- TNBC:
-
Triple-negative breast cancer
- TTP:
-
Time to progression
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424.
Soysal SD, Tzankov A, Muenst SE. Role of the tumor microenvironment in breast cancer. Pathobiology. 2015;82(3–4):142–52.
Hanahan D, Coussens LM. Accessories to the crime: functions of cells recruited to the tumor microenvironment. Cancer Cell. 2012;21(3):309–22.
Loi S, Drubay D, Adams S, Pruneri G, Francis PA, Lacroix-Triki M, et al. Tumor-infiltrating lymphocytes and prognosis: a pooled individual patient analysis of early-stage triple-negative breast cancers. J Clin Oncol. 2019;37(7):559–69.
Salgado R, Denkert C, Demaria S, Sirtaine N, Klauschen F, Pruneri G, et al. The evaluation of tumor-infiltrating lymphocytes (TILs) in breast cancer: recommendations by an International TILs Working Group 2014. Ann Oncol. 2015;26(2):259–71.
Badr NM, Berditchevski F, Shaaban AM. The immune microenvironment in breast carcinoma: predictive and prognostic role in the neoadjuvant setting. Pathobiology. 2020;87(2):61–74.
de la Cruz-Merino L, Barco-Sánchez A, Henao Carrasco F, Nogales Fernández E, Vallejo Benítez A, Brugal Molina J, et al. New insights into the role of the immune microenvironment in breast carcinoma. Clin Dev Immunol. 2013;2013: 785317.
Ran S, Volk L, Hall K, Flister MJ. Lymphangiogenesis and lymphatic metastasis in breast cancer. Pathophysiology. 2010;17(4):229–51.
Howlader N, Noone AM, Krapcho M, Miller D, Brest A, Yu M, et al. SEER Cancer Statistics Review, 1975–2017. In: National Cancer Institute. 2020. https://seer.cancer.gov/csr/1975_2017/. Accessed 27 Jul 2021.
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70(1):7–30.
Colzani E, Johansson ALV, Liljegren A, Foukakis T, Clements M, Adolfsson J, et al. Time-dependent risk of developing distant metastasis in breast cancer patients according to treatment, age and tumour characteristics. Br J Cancer. 2014;110(5):1378–84.
Gibert-Ramos A, López C, Bosch R, Fontoura L, Bueno G, García-Rojo M, et al. Immune response profile of primary tumour, sentinel and non-sentinel axillary lymph nodes related to metastasis in breast cancer: an immunohistochemical point of view. Histochem Cell Biol. 2019;152(3):177–93.
Chang AY, Bhattacharya N, Mu J, Setiadi AF, Carcamo-Cavazos V, Lee GH, et al. Spatial organization of dendritic cells within tumor draining lymph nodes impacts clinical outcome in breast cancer patients. J Transl Med. 2013;11:242.
Faghih Z, Erfani N, Haghshenas MR, Safaei A, Talei AR, Ghaderi A. Immune profiles of CD4+ lymphocyte subsets in breast cancer tumor draining lymph nodes. Immunol Lett. 2014;158(1–2):57–65.
Li M, Li A, Zhou S, Xu Y, Xiao Y, Bi R, et al. Heterogeneity of PD-L1 expression in primary tumors and paired lymph node metastases of triple negative breast cancer. BMC Cancer. 2018;18(1):4.
Kohrt HE, Nouri N, Nowels K, Johnson D, Holmes S, Lee PP. Profile of immune cells in axillary lymph nodes predicts disease-free survival in breast cancer. PLoS Med. 2005;2(9): e284.
López C, Bosch R, Orero G, Korzynska A, García-Rojo M, Bueno G, et al. The immune response in nonmetastatic axillary lymph nodes is associated with the presence of axillary metastasis and breast cancer patient outcome. Am J Pathol. 2020;190(3):660–73.
López C, Gibert-Ramos A, Bosch R, Korzynska A, García-Rojo M, Bueno G, et al. Differences in the immune response of the nonmetastatic axillary lymph nodes between triple-negative and luminal a breast cancer surrogate subtypes. Am J Pathol. 2021;191(3):545–54.
Li X, Yang J, Peng L, Sahin AA, Huo L, Ward KC, et al. Triple-negative breast cancer has worse overall survival and cause-specific survival than non-triple-negative breast cancer. Breast Cancer Res Treat. 2017;161(2):279–87.
Howlader N, Cronin KA, Kurian AW, Andridge R. Differences in breast cancer survival by molecular subtypes in the United States. Cancer Epidemiol Biomarkers Prev. 2018;27(6):619–26.
WHO Classification of Tumours Editorial Board. Breast Tumours. WHO Classification of Tumours series, vol. 2. 5th ed. Lyon: International Agency for Research on Cancer; 2019.
Vittinghoff E, McCulloch CE. Relaxing the rule of ten events per variable in logistic and Cox regression. Am J Epidemiol. 2007;165(6):710–8.
Callau C, Lejeune M, Korzynska A, García M, Bueno G, Bosch R, et al. Evaluation of cytokeratin-19 in breast cancer tissue samples: a comparison of automatic and manual evaluations of scanned tissue microarray cylinders. Biomed Eng Online. 2015;14(Suppl 2):S2.
Pelekanou V, Villarroel-Espindola F, Schalper KA, Pusztai L, Rimm DL. CD68, CD163, and matrix metalloproteinase 9 (MMP-9) co-localization in breast tumor microenvironment predicts survival differently in ER-positive and -negative cancers. Breast Cancer Res. 2018;20(1):154.
Pinder SE, Brown JP, Gillett C, Purdie CA, Speirs V, Thompson AM, et al. The manufacture and assessment of tissue microarrays: suggestions and criteria for analysis, with breast cancer as an example. J Clin Pathol. 2013;66(3):169–77.
Fernández-Carrobles MDM, Bueno G, Déniz O, Salido J, García-Rojo M. Automatic handling of tissue microarray cores in high-dimensional microscopy images. IEEE J Biomed Health Inform. 2014;18(3):999–1007.
Roszkowiak L, Lopez C. PATMA: parser of archival tissue microarray. PeerJ. 2016;4: e2741.
López C, Lejeune M, Bosch R, Korzynska A, García-Rojo M, Salvadó MT, et al. Digital image analysis in breast cancer: an example of an automated methodology and the effects of image compression. Stud Health Technol Inform. 2012;179:155–71.
Korzynska A, Roszkowiak L, Zak J, Lejeune M, Orero G, Bosch R, et al. The METINUS Plus method for nuclei quantification in tissue microarrays of breast cancer and axillary node tissue section. Biomed Signal Process Control. 2017;32:1–9.
DeLong ER, DeLong DM, Clarke-Pearson DL. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics. 1988;44(3):837–45.
Loi S, Sirtaine N, Piette F, Salgado R, Viale G, Van Eenoo F, et al. Prognostic and predictive value of tumor-infiltrating lymphocytes in a phase III randomized adjuvant breast cancer trial in node-positive breast cancer comparing the addition of docetaxel to doxorubicin with doxorubicin-based chemotherapy: BIG 02–98. J Clin Oncol. 2013;31(7):860–7.
Tian Z, Tang J, Liao X, Yang Q, Wu Y, Wu G. An immune-related prognostic signature for predicting breast cancer recurrence. Cancer Med. 2020;9(20):7672–85.
Yu S, Hu C, Liu L, Cai L, Du X, Yu Q, et al. Comprehensive analysis and establishment of a prediction model of alternative splicing events reveal the prognostic predictor and immune microenvironment signatures in triple negative breast cancer. J Transl Med. 2020;18(1):286.
Iwamoto M, Shinohara H, Miyamoto A, Okuzawa M, Mabuchi H, Nohara T, et al. Prognostic value of tumor-infiltrating dendritic cells expressing CD83 in human breast carcinomas. Int J Cancer. 2003;104(1):92–7.
Ni C, Yang L, Xu Q, Yuan H, Wang W, Xia W, et al. CD68- and CD163-positive tumor infiltrating macrophages in non-metastatic breast cancer: a retrospective study and meta-analysis. J Cancer. 2019;10(19):4463–72.
Yuan ZY, Luo RZ, Peng RJ, Wang SS, Xue C. High infiltration of tumor-associated macrophages in triple-negative breast cancer is associated with a higher risk of distant metastasis. Onco Targets Ther. 2014;7:1475–80.
Hu G, Wang S. Prognostic role of tumor-infiltrating CD57-positive lymphocytes in solid tumors: a meta-analysis. Oncotarget. 2018;9(8):8111–9.
Peng GL, Li L, Guo YW, Yu P, Yin XJ, Wang S, et al. CD8(+) cytotoxic and FoxP3(+) regulatory T lymphocytes serve as prognostic factors in breast cancer. Am J Transl Res. 2019;11(8):5039–53.
Liu S, Lachapelle J, Leung S, Gao D, Foulkes WD, Nielsen TO. CD8+ lymphocyte infiltration is an independent favorable prognostic indicator in basal-like breast cancer. Breast Cancer Res. 2012;14(2):R48.
Aerts-Toegaert C, Heirman C, Tuyaerts S, Corthals J, Aerts JL, Bonehill A, et al. CD83 expression on dendritic cells and T cells: correlation with effective immune responses. Eur J Immunol. 2007;37(3):686–95.
Poindexter NJ, Sahin A, Hunt KK, Grimm EA. Analysis of dendritic cells in tumor-free and tumor-containing sentinel lymph nodes from patients with breast cancer. Breast Cancer Res. 2004;6(4):R408–15.
Kichler-Lakomy C, Budinsky AC, Wolfram R, Hellan M, Wiltschke C, Brodowicz T, et al. Deficiences in phenotype expression and function of dentritic cells from patients with early breast cancer. Eur J Med Res. 2006;11(1):7–12.
Kashimura S, Saze Z, Terashima M, Soeta N, Ohtani S, Osuka F, et al. CD83(+) dendritic cells and Foxp3(+) regulatory T cells in primary lesions and regional lymph nodes are inversely correlated with prognosis of gastric cancer. Gastric Cancer. 2012;15(2):144–53.
Furihata M, Ono Y, Ichikawa K, Tomita S, Fujimori T, Kubota K. Prognostic significance of CD83 positive, mature dendritic cells in the gallbladder carcinoma. Oncol Rep. 2005;14(2):353–6.
Wculek SK, Cueto FJ, Mujal AM, Melero I, Krummel MF, Sancho D. Dendritic cells in cancer immunology and immunotherapy. Nat Rev Immunol. 2020;20(1):7–24.
Treilleux I, Blay JY, Bendriss-Vermare N, Ray-Coquard I, Bachelot T, Guastalla JP, et al. Dendritic cell infiltration and prognosis of early stage breast cancer. Clin Cancer Res. 2004;10(22):7466–74.
Mansfield AS, Heikkila P, von Smitten K, Vakkila J, Leidenius M. Metastasis to sentinel lymph nodes in breast cancer is associated with maturation arrest of dendritic cells and poor co-localization of dendritic cells and CD8+ T cells. Virchows Arch. 2011;459(4):391–8.
La Rocca G, Anzalone R, Corrao S, Magno F, Rappa F, Marasà S, et al. CD1a down-regulation in primary invasive ductal breast carcinoma may predict regional lymph node invasion and patient outcome. Histopathology. 2008;52(2):203–12.
Kanao H, Enomoto T, Kimura T, Fujita M, Nakashima R, Ueda Y, et al. Overexpression of LAMP3/TSC403/DC-LAMP promotes metastasis in uterine cervical cancer. Cancer Res. 2005;65(19):8640–5.
Ishigami S, Ueno S, Matsumoto M, Okumura H, Arigami T, Uchikado Y, et al. Prognostic value of CD208-positive cell infiltration in gastric cancer. Cancer Immunol Immunother. 2010;59(3):389–95.
The Lancet Oncology. Calling time on the immunotherapy gold rush. Lancet Oncol. 2017;18(8):981.
Acknowledgements
We thank Barbara Tomàs, María del Mar Barbera, Noelia Burgues, Ainhoa Montserrat, Almudena Nagera, Montse Salvadó, Ana Suñé, Beatriu Domenech, Anna Guasch, Marita Curto, and Marc Iniesta for their skilful technical assistance.
Funding
Carlos López is the guarantor of this work and, as such, had full access to all the data in the study. He takes responsibility for the integrity of the data and the accuracy of the data analysis. This work was funded by projects PI11/0488 (Principal Investigator: C.L.) and PI13/02501 (Principal investigator: M.L.) of the Institute of Health Carlos III, which is the main public research body that funds, manages and carries out biomedical research in Spain. It was co-funded by the European Union European Regional Development Fund. It was also supported by the Project AIDPATH FP7-PEOPLE Project ID: 612471 (Principal Investigator: G.B.). The biopsies were selected from the Banc de Tumors of the HTVC, member of the Xarxa de Bancs de Tumors de Catalunya (XBTC), sponsored by the Oncology Master Plan for Catalonia (Pla Director d’Oncologia de Catalunya).
Author information
Authors and Affiliations
Contributions
CL, RB, DM and ML designed the study and selected the sample. CL, JFGF, SMG, JCR, MA, JG, MB, SRV, MLL and ML collected data and reviewed the clinical records. CL, ESC, JCR and ML processed the samples. CL, AK, MGR, GB, LR and JB digitized the samples and developed image analysis procedures. CL, AGN, ESC, AR and ML did the statistical analysis. CL, AGN, ESC and ML designed the figures. All the authors have read and reviewed the article and made significant contributions to the interpretation of the data.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Ethics approval and consent to participate
This study was approved by the Ethics Committee of Hospital Joan XXIII de Tarragona (Reference number: 24p/2012) and by the Research Committee of the HTVC. All patients provided their written informed consent to participate in the study and for the use of their biopsy tissues and clinical data, in accordance with Spanish law. We followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
Consent for publication
Yes.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
López, C., Bosch, R., Korzynska, A. et al. CD68 and CD83 immune populations in non-metastatic axillary lymph nodes are of prognostic value for the survival and relapse of breast cancer patients. Breast Cancer 29, 618–635 (2022). https://doi.org/10.1007/s12282-022-01336-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12282-022-01336-2