Abstract
Objective
To determine whether sarcopenia can potentially predict worse survival after resection of pancreatic ductal adenocarcinoma.
Background
Sarcopenia is correlated with poor outcomes in hepatopancreatobiliary malignancies, but the relationship of both its qualitative and quantitative features with patient survival after pancreatectomy has not been investigated in a western population.
Patients and Methods
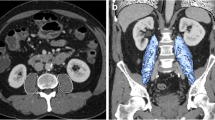
Preoperative cross-sectional computed tomography scans of consecutive patients who underwent pancreatectomy in 2005–2017 were evaluated for skeletal muscle index (SMI), intramuscular adipose tissue content (IMAC), and visceral-to-subcutaneous adipose tissue area ratio (VSR). Sex-specific categorical cut-offs were determined. Findings were correlated with outcome.
Results
The study included 111 patients, 47% of whom were female, with a median age of 67 years (range: 35–87 years), and median body mass index of 23 kg/m2 (range: 16–40 kg/m2); 77% had a Whipple procedure and 66% received adjuvant chemotherapy. Low SMI correlated with poor overall survival (OS) (P = 0.007), disease-specific survival (DSS) (P = 0.006), and recurrence-free survival (RFS) (P = 0.01). High IMAC correlated with poor OS (P = 0.04). Patients with high IMAC tended to have a shorter DSS (P = 0.09), with no correlation with RFS (P = 0.6). VSR was not associated with survival. Multivariable analysis yielded an independent association of low SMI with OS (HR = 1.7, 95%CI: 1.1–2.8, P = 0.02), DSS (HR = 1.8, 95%CI: 1.03–3.2, P = 0.04), and RFS (HR = 1.8, 95%CI: 1.1–2.8, P = 0.01), and of high IMAC with OS (HR = 1.9, 95%CI: 1.1–3.1, P = 0.01).
Conclusion
Both qualitative and quantitative measures of skeletal muscle were independently associated with impaired survival in patients with resectable PDAC. Sarcopenia might serve as an early radiographic surrogate of aggressive tumor behavior, with potential implications for clinical decision-making and future study.



Similar content being viewed by others
References
Schucht P, Fischer U, Fung C, et al. Follow-up computed tomography after evacuation of chronic subdural hematoma. N Engl J Med. 2019;380(12):1186–7. https://doi.org/10.1056/NEJMc1812507.
McGuigan A, Kelly P, Turkington RC, Jones C, Coleman HG, McCain RS. Pancreatic cancer: a review of clinical diagnosis, epidemiology, treatment and outcomes. World J Gastroenterol. 2018;24(43):4846–61. https://doi.org/10.3748/wjg.v24.i43.4846.
Khorana AA, Shapiro M, Mangu PB, et al. Potentially curable pancreatic cancer: American society of clinical oncology clinical practice guideline update. J Clin Oncol. 2017;35(20):2324–8. https://doi.org/10.1200/JCO.2017.72.4948.
Reddy S, Wolfgang CL, Cameron JL, et al. Total pancreatectomy for pancreatic adenocarcinoma: evaluation of morbidity and long-term Survival. Ann Surg. 2009;250(2):282–7. https://doi.org/10.1097/SLA.0b013e3181ae9f93.
Kneuertz PJ, Pitt HA, Bilimoria KY, et al. Risk of morbidity and mortality following hepato-pancreato-biliary surgery. J Gastrointest Surg. 2012;16(9):1727–35. https://doi.org/10.1007/s11605-012-1938-y.
Pulvirenti A, Pea A, Rezaee N, et al. Perioperative outcomes and long-term quality of life after total pancreatectomy. Br J Surg. 2019;106(13):1819–28. https://doi.org/10.1002/bjs.11185.
Rahib L, Smith BD, Aizenberg R, Rosenzweig AB, Fleshman JM, Matrisian LM. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the united states. Cancer Res. 2014;74(11):2913–21. https://doi.org/10.1158/0008-5472.CAN-14-0155.
Yamada S, Fujii T, Sugimoto H, et al. Aggressive surgery for borderline resectable pancreatic cancer. Pancreas. 2013;42(6):1004–10. https://doi.org/10.1097/mpa.0b013e31827b2d7c.
Gurusamy KS, Kumar S, Davidson BR, Fusai G. Resection versus other treatments for locally advanced pancreatic cancer. Cochrane Database Syst Rev. 2014. https://doi.org/10.1002/14651858.CD010244.pub2.
Chindapasirt J. Sarcopenia in cancer patients. Asian Pac J Cancer Prev. 2016;16(18):8075–7. https://doi.org/10.7314/APJCP.2015.16.18.8075.
Ryan AM, Power DG, Daly L, Cushen SJ, Ní Bhuachalla E, Prado CM. Cancer-associated malnutrition, cachexia and sarcopenia: the skeleton in the hospital closet 40 years later. Proc Nutr Soc. 2016;75(2):199–211. https://doi.org/10.1017/S002966511500419X.
Peterson SJ, Mozer M. Differentiating sarcopenia and cachexia among patients with cancer. Nutr Clin Pract. 2017;32(1):30–9. https://doi.org/10.1177/0884533616680354.
Sheetz KH, Waits SA, Terjimanian MN, et al. Cost of major surgery in the sarcopenic patient. J Am Coll Surg. 2013;217(5):813–8. https://doi.org/10.1016/j.jamcollsurg.2013.04.042.
Gani F, Buettner S, Margonis GA, et al. Sarcopenia predicts costs among patients undergoing major abdominal operations. Surg (US). 2016;160(5):1162–71. https://doi.org/10.1016/j.surg.2016.05.002.
Friedman J, Lussiez A, Sullivan J, Wang S, Englesbe M. Implications of sarcopenia in major surgery. Nutr Clin Pract. 2015;30(2):175–9. https://doi.org/10.1177/0884533615569888.
Ida S, Watanabe M, Yoshida N, et al. Sarcopenia is a predictor of postoperative respiratory complications in patients with esophageal cancer. Ann Surg Oncol. 2015;22(13):4432–7. https://doi.org/10.1245/s10434-015-4559-3.
Joglekar S, Nau PN, Mezhir JJ. The impact of sarcopenia on survival and complications in surgical oncology: a review of the current literature. J Surg Oncol. 2015;112(5):503–9. https://doi.org/10.1002/jso.24025.
Kuwada K, Kuroda S, Kikuchi S, et al. Sarcopenia and comorbidity in gastric cancer surgery as a useful combined factor to predict eventual death from other causes. Ann Surg Oncol. 2018;25(5):1160–6. https://doi.org/10.1245/s10434-018-6354-4.
Simonsen C, De Heer P, Bjerre ED, et al. Sarcopenia and postoperative complication risk in gastrointestinal surgical oncology. Ann Surg. 2018;268(1):58–69. https://doi.org/10.1097/SLA.0000000000002679.
Dolan DR, Knight KA, Maguire S, Moug SJ. The relationship between sarcopenia and survival at 1 year in patients having elective colorectal cancer surgery. Tech Coloproctol. 2019;23(9):877–85. https://doi.org/10.1007/s10151-019-02072-0.
Levolger S, Van Vugt JLA, De Bruin RWF, IJzermans JNM,. Systematic review of sarcopenia in patients operated on for gastrointestinal and hepatopancreatobiliary malignancies. Br J Surg. 2015;102(12):1448–58. https://doi.org/10.1002/bjs.9893.
Peng P, Hyder O, Firoozmand A, et al. Impact of sarcopenia on outcomes following resection of pancreatic adenocarcinoma. J Gastrointest Surg. 2012;16(8):1478–86. https://doi.org/10.1007/s11605-012-1923-5.
Tan BHL, Birdsell LA, Martin L, Baracos VE, Fearon KCH. Sarcopenia in an overweight or obese patient is an adverse prognostic factor in pancreatic cancer. Clin Cancer Res. 2009;15(22):6973–9. https://doi.org/10.1158/1078-0432.CCR-09-1525.
Park J, Han S-S, Park SJ, Park SW. Risk factors affecting incidence of morbidity after pancreaticoduodenectomy. HPB. 2018;20:627. https://doi.org/10.1016/j.hpb.2018.06.2204.
Hile E, Hoffman L, Postier R, Ding K, Yang J, Li M. A pilot RCT of sarcopenia-focused prehabilitation in pancreas cancer. J Clin Oncol. 2017;35(15):TPS10127. https://doi.org/10.1200/JCO.2017.35.15_suppl.TPS10127.
Van Vledder MG, Levolger S, Ayez N, Verhoef C, Tran TCK, Ijzermans JNM. Body composition and outcome in patients undergoing resection of colorectal liver metastases. Br J Surg. 2012;99(4):550–7. https://doi.org/10.1002/bjs.7823.
Shen W, Punyanitya M, Wang ZM, et al. Total body skeletal muscle and adipose tissue volumes: estimation from a single abdominal cross-sectional image. J Appl Physiol. 2004;68(6):2333–8. https://doi.org/10.1016/j.jhep.2017.11.030.
van Vugt JLA, Alferink LJM, Buettner S, et al. A model including sarcopenia surpasses the MELD score in predicting waiting list mortality in cirrhotic liver transplant candidates: a competing risk analysis in a national cohort. J Hepatol. 2017;68(4):707–14. https://doi.org/10.1016/j.jhep.2017.11.030.
Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240(2):205–13. https://doi.org/10.1097/01.sla.0000133083.54934.ae.
Chun YS, Pawlik TM, Vauthey JN. 8th Edition of the AJCC cancer staging manual: pancreas and hepatobiliary cancers. Ann Surg Oncol. 2018;25(4):845–7. https://doi.org/10.1245/s10434-017-6025-x.
Verbeke CS, Leitch D, Menon KV, McMahon MJ, Guillou PJ, Anthoney A. Redefining the R1 resection in pancreatic cancer. Br J Surg. 2006;93(10):1232–7. https://doi.org/10.1002/bjs.5397.
Esposito I, Kleeff J, Bergmann F, et al. Most pancreatic cancer resections are R1 resections. Ann Surg Oncol. 2008;15(6):1651–60. https://doi.org/10.1245/s10434-008-9839-8.
Okumura S, Kaido T, Hamaguchi Y, et al. Impact of preoperative quality as well as quantity of skeletal muscle on survival after resection of pancreatic cancer. Surg (US). 2015;157(6):1088–98. https://doi.org/10.1016/j.surg.2015.02.002.
Sugimoto M, Farnell MB, Nagorney DM, et al. Decreased skeletal muscle volume is a predictive factor for poorer survival in patients undergoing surgical resection for pancreatic ductal adenocarcinoma. J Gastrointest Surg. 2018;I:831–9.
Holly EA, Chaliha I, Bracci PM, Gautam M. Signs and symptoms of pancreatic cancer: a population-based case-control study in the San Francisco Bay area. Clin Gastroenterol Hepatol. 2004;2(6):510–7. https://doi.org/10.1016/S1542-3565(04)00171-5.
Fearon K, Strasser F, Anker SD, et al. Definition and classification of cancer cachexia: an international consensus. Lancet Oncol. 2011;12(5):489–95. https://doi.org/10.1016/S1470-2045(10)70218-7.
Sah RP, Sharma A, Nagpal S, et al. Phases of metabolic and soft tissue changes in months preceding a diagnosis of pancreatic ductal adenocarcinoma. Gastroenterology. 2019;156(6):1742–52. https://doi.org/10.1053/j.gastro.2019.01.039.
Hart PA, Kamada P, Rabe KG, et al. Weight loss precedes cancer-specific symptoms in pancreatic cancer-associated diabetes mellitus. Pancreas. 2011;40(5):768–72. https://doi.org/10.1097/MPA.0b013e318220816a.
Danai LV, Babic A, Rosenthal MH, et al. Altered exocrine function can drive adipose wasting in early pancreatic cancer. Nature. 2018;558:600–4. https://doi.org/10.1038/s41586-018-0235-7.
Javed AA, Wright MJ, Siddique A, et al. Outcome of patients with borderline resectable pancreatic cancer in the contemporary era of neoadjuvant chemotherapy. J Gastrointest Surg. 2019;23(1):112–21. https://doi.org/10.1007/s11605-018-3966-8.
Ahmad SA, Duong M, Sohal DPS, et al. Surgical outcome results from SWOG S1505: a randomized clinical trial of mFOLFIRINOX versus gemcitabine/nab-paclitaxel for perioperative treatment of resectable pancreatic ductal adenocarcinoma. Ann Surg. 2020;272(3):481–6. https://doi.org/10.1097/SLA.0000000000004155.
Sohal DPS, Duong M, Ahmad SA, et al. Efficacy of perioperative chemotherapy for resectable pancreatic adenocarcinoma: a phase 2 randomized clinical trial. JAMA Oncol. 2021;7(3):421–7. https://doi.org/10.1001/jamaoncol.2020.7328.
National Comprehensive Cancer Network (NCCN). Pancreatic adenocarcinoma (version 2.2021). https://www.nccn.org/professionals/physician_gls/pdf/pancreatic.pdf. Published 2021. Accessed from 3 Sep 2021.
Funding
No funding was procured for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
There are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rom, H., Tamir, S., Van Vugt, J.L.A. et al. Sarcopenia as a Predictor of Survival in Patients with Pancreatic Adenocarcinoma After Pancreatectomy. Ann Surg Oncol 29, 1553–1563 (2022). https://doi.org/10.1245/s10434-021-10995-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-021-10995-y