Abstract
This paper studies the social acceptability and feasibility of “herd immunity” strategies against coronavirus disease 2019 (COVID-19). To this end, we propose a control scheme that aims to develop herd immunity while satisfying the following two basic requirements for a viable policy option. The first requirement is social acceptability: the overall deaths should be minimized for social acceptance. The second is feasibility: the healthcare system should not be overwhelmed to avoid diverse adverse effects. Exploiting the fact that the severity of the disease increases considerably with age, the proposed control scheme protects high-risk individuals that mainly consist of the elderly. The protection of high-risk individuals reduces the average severity of the disease in the unprotected population, thereby substantially reducing mortality and avoiding the collapse of the healthcare system. Social acceptability (in terms of the resulting mortality) and feasibility (in terms of the healthcare system capacity) of the proposed herd immunity strategy are summarized into two respective, easily computable conditions by building on a simple susceptible–infected–recovered (SIR) model. For its parsimony, the proposed framework can provide a rule of thumb that applies to various populations for studying the viability of herd immunity strategies against COVID-19. For Japan, herd immunity may be developed by the considered control scheme if 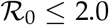 and the severity rates of the disease are 1/10 times smaller than the previously reported value, although as high mortality as seasonal influenza is expected.
and the severity rates of the disease are 1/10 times smaller than the previously reported value, although as high mortality as seasonal influenza is expected.
Competing Interest Statement
The authors have declared no competing interest.
Funding Statement
No funding sources were used.
Author Declarations
I confirm all relevant ethical guidelines have been followed, and any necessary IRB and/or ethics committee approvals have been obtained.
Yes
All necessary patient/participant consent has been obtained and the appropriate institutional forms have been archived.
Yes
I understand that all clinical trials and any other prospective interventional studies must be registered with an ICMJE-approved registry, such as ClinicalTrials.gov. I confirm that any such study reported in the manuscript has been registered and the trial registration ID is provided (note: if posting a prospective study registered retrospectively, please provide a statement in the trial ID field explaining why the study was not registered in advance).
Yes
I have followed all appropriate research reporting guidelines and uploaded the relevant EQUATOR Network research reporting checklist(s) and other pertinent material as supplementary files, if applicable.
Yes
Data Availability
All the data used in the manuscript are publicly available.






