Abstract
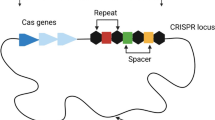
The CRISPR–Cas are adaptive immune systems found in archaea and bacteria, responsible for providing sequence-specific resistance against foreign DNA. Strains of Pseudomonas aeruginosa may carry CRISPR/Cas system types I-F, I-E and/or I-C; however, several aspects related to the epidemiology and functionality of these systems have not yet been revealed. Here, we report 13 genomes of clinical strains of P. aeruginosa from Brazil that were positive for CRISPR/Cas system types I-F and I-E, a rare feature in this species. The phylogenetic tree, which was constructed with 161 other publicly available genomes, suggested no direct relationship between positive strains, and the various types of CRISPR/Cas systems were spread throughout the tree. Comparative analysis of the genetic locations of type I-F and a specific orphan CRISPR array (without cas genes), named the LES locus, showed sequence similarities between this orphan locus and type I-F, but these LES loci were inserted in a different genomic location. We also report the presence of prophages, the presence of anti-CRISPR genes, and possibly the presence of self-targeting spacers. Here, we conclude that CRISPR/Cas is highly associated with certain lineages and is spread throughout the phylogenetic tree, showing no clear pattern of evolutionary distribution. Moreover, the LES locus might be a CRISPR1 locus related to type I-F that may have been misplaced and maintained over time. Furthermore, strains carrying I-F and I-E are rare, and not all of them are closely related. Further functional work is needed to better comprehend if aspects reported in this study are functional, including the LES locus, self-targeting spacers, anti-CRISPR protection, and I-F/I-E-carrying lineages.



Similar content being viewed by others
Availability of data and material
The BioProject has been deposited at the GenBank database under the accession number PRJNA514718.
References
Andrews S (2010) FastQC: A quality control tool for high throughput sequence data. Babraham Bioinformatics. https://www.bioinformatics.babraham.ac.uk/projects/fastqc/
Arndt D, Grant JR, Marcu A et al (2016) PHASTER: a better, faster version of the PHAST phage search tool. Nucleic Acids Res 44:W16–W21. https://doi.org/10.1093/nar/gkw387
Azam MW, Khan AU (2019) Updates on the pathogenicity status of Pseudomonas aeruginosa. Drug Discov Today 00:1–10. https://doi.org/10.1016/j.drudis.2018.07.003
Bankevich A, Nurk S, Antipov D et al (2012) SPAdes: a new genome assembly algorithm and its applications to single-cell sequencing. J Comput Biol 19:455–477. https://doi.org/10.1089/cmb.2012.0021
Berriman M, Rutherford K (2003) Viewing and annotating sequences in Artemis. Brief Bioinform 4:124–132
Biswas A, Gagnon JN, Brouns SJJJ et al (2013) CRISPRTarget - Bioinformatic prediction and analysis of crRNA targets. RNA Biol 10:817–827. https://doi.org/10.4161/rna.24046
Cady KC, O’Toole GA (2011) Non-identity-mediated CRISPR-bacteriophage interaction mediated via the Csy and Cas3 proteins. J Bacteriol 193:3433–3445. https://doi.org/10.1128/JB.01411-10
Castresana J (2000) Selection of conserved blocks from multiple alignments for their use in phylogenetic analysis. Mol Biol Evol 17:540–552
Couvin D, Bernheim A, Toffano-Nioche C et al (2018) CRISPRCasFinder, an update of CRISRFinder, includes a portable version, enhanced performance and integrates search for Cas proteins. Nucleic Acids Res 46:W246–W251. https://doi.org/10.1093/nar/gky425
de Luz AC, O, da Silva JMA, Rezende AM, et al (2019) Analysis of direct repeats and spacers of CRISPR/Cas systems type I-F in Brazilian clinical strains of Pseudomonas aeruginosa. Mol Genet Genomics. https://doi.org/10.1007/s00438-019-01575-7
England WE, Kim T, Whitaker RJ (2018) Metapopulation Structure of CRISPR-Cas Immunity in Pseudomonas aeruginosa and Its Viruses. mSystems 3: e00075–18
Essoh C, Blouin Y, Loukou G et al (2013) The susceptibility of Pseudomonas aeruginosa strains from cystic fibrosis patients to bacteriophages. PLoS ONE. https://doi.org/10.1371/journal.pone.0060575
Francisco AP, Bugalho M, Ramirez M, Carriço JA (2009) Global optimal eBURST analysis of multilocus typing data using a graphic matroid approach. BMC Bioinformatics 10:1–15. https://doi.org/10.1186/1471-2105-10-152
Giske CG, Libisch B, Colinon C et al (2006) Establishing clonal relationships between VIM-1-like metallo-β- lactamase-producing Pseudomonas aeruginosa strains from four European countries by multilocus sequence typing. J Clin Microbiol 44:4309–4315. https://doi.org/10.1128/JCM.00817-06
Gurevich A, Saveliev V, Vyahhi N, Tesler G (2013) QUAST: Quality assessment tool for genome assemblies. Bioinformatics 29:1072–1075. https://doi.org/10.1093/bioinformatics/btt086
Hille F, Richter H, Wong SP et al (2018) The Biology of CRISPR-Cas: Backward and Forward. Cell 172:1239–1259. https://doi.org/10.1016/j.cell.2017.11.032
Irvine S, Bunk B, Bayes HK et al (2019) Genomic and transcriptomic characterization of Pseudomonas aeruginosa small colony variants derived from a chronic infection model. Microb Genomics 5:1–11. https://doi.org/10.1099/mgen.0.000262
Johnson M, Zaretskaya I, Raytselis Y et al (2008) NCBI BLAST: a better web interface. Nucleic Acids Res 36:5–9. https://doi.org/10.1093/nar/gkn201
Jolley KA, Bray JE, Maiden MCJ (2018) Open-access bacterial population genomics: BIGSdb software, the PubMLST.org website and their applications. Wellcome Open Res. 3:124. https://doi.org/10.12688/wellcomeopenres.14826.1
Joshi NA, Fass JN (2011) Sickle: A sliding-window, adaptive, quality-based trimming tool for FastQ files (Version 1.33) [Software]. https://github.com/najoshi/sickle.
Katoh K, Standley DM (2013) MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Mol Biol Evol 30:772–780. https://doi.org/10.1093/molbev/mst010
Koonin EV, Makarova KS, Zhang F (2017) Diversity, classification and evolution of CRISPR-Cas systems. Curr Opin Microbiol 37:67–78. https://doi.org/10.1016/j.mib.2017.05.008
Letunic I, Bork P (2019) Interactive Tree Of Life (iTOL) v4: recent updates and new developments. Nucleic Acids Res 47:W256–W259. https://doi.org/10.1093/nar/gkz239
Martin M (2013) Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J 17:10–12. https://doi.org/10.14806/ej.17.1.200
Minh BQ, Schmidt HA, Chernomor O et al (2020) IQ-TREE 2: new models and efficient methods for phylogenetic inference in the genomic era. Mol Biol Evol 37:1530–1534. https://doi.org/10.1093/molbev/msaa015
Nylander JAA (2004) MrModeltest v2. Program distributed by the author. Evolutionary Biology Centre, Uppsala University. https://github.com/nylander/MrModeltest2
Parkins MD, Somayaji R, Waters J (2018) Epidemiology, Biology, and Impact of Clonal Pseudomonas aeruginosa Infections in Cystic Fibrosis. Clin Microbiol Rev 31: 1–38. cystic fibrosis. Clin Microbiol Rev 31:e00019–18.https://doi.org/https://doi.org/10.1128/CMR.00019-18.
Pawluk A, Davidson AR, Maxwell KL (2018) Anti-CRISPR: discovery, mechanism and function. Nat Rev Microbiol 16:12–17. https://doi.org/10.1038/nrmicro.2017.120
Pawluk A, Staals RHJJ, Taylor C et al (2016) Inactivation of CRISPR-Cas systems by anti-CRISPR proteins in diverse bacterial species. Nat Microbiol 1:1–6. https://doi.org/10.1038/nmicrobiol.2016.85
Pereira RPA, Peplies J, Mushi D et al (2018) Pseudomonas-Specific NGS Assay Provides Insight Into Abundance and Dynamics of Pseudomonas Species Including P. aeruginosa in a Cooling Tower. Front Microbiol 9:1–15. https://doi.org/10.3389/fmicb.2018.01958
Pundir S, Martin MJ, O’Donovan C, Consortium TU (2016) UniProt Tools. Curr Protoc Bioinforma 53:1.29.1–1.29.15. doi: https://doi.org/10.1002/0471250953.bi0129s53
Ronquist F, Teslenko M, van der Mark P et al (2012) MrBayes 3.2: efficient bayesian phylogenetic inference and model choice across a large model space. Syst Biol 61:539–542. https://doi.org/10.1093/sysbio/sys029
Seemann T (2014) Prokka: Rapid prokaryotic genome annotation. Bioinformatics 30:2068–2069. https://doi.org/10.1093/bioinformatics/btu153
Sela I, Ashkenazy H, Katoh K, Pupko T (2015) GUIDANCE2: Accurate detection of unreliable alignment regions accounting for the uncertainty of multiple parameters. Nucleic Acids Res 43:W7–W14. https://doi.org/10.1093/nar/gkv318
Silby MW, Winstanley C, Godfrey SAC et al (2011) Pseudomonas genomes: Diverse and adaptable. FEMS Microbiol Rev 35:652–680. https://doi.org/10.1111/j.1574-6976.2011.00269.x
Silveira MC, Rocha-De-Souza CM, Albano RM et al (2020) Exploring the success of Brazilian endemic clone Pseudomonas aeruginosa ST277 and its association with the CRISPR-Cas system type I-C. BMC Genomics 21:4–11. https://doi.org/10.1186/s12864-020-6650-9
Stern A, Keren L, Wurtzel O et al (2010) Self-targeting by CRISPR: Gene regulation or autoimmunity? Trends Genet 26:335–340. https://doi.org/10.1016/j.tig.2010.05.008
Swofford D (2002). PAUP*. Phylogenetic Analysis Using Parsimony (*and Other Methods) (Version 4) [Software]. https://paup.phylosolutions.com/
Valot B, Guyeux C, Rolland JY et al (2015) What it takes to be a Pseudomonas aeruginosa? The core genome of the opportunistic pathogen updated. PLoS ONE 10:1–15. https://doi.org/10.1371/journal.pone.0126468
van Belkum A, Soriaga LB, LaFave MC et al (2015) Phylogenetic distribution of CRISPR-Cas systems in antibiotic-resistant Pseudomonas aeruginosa. MBio 6:1–13. https://doi.org/10.1128/mBio.01796-15
van der Oost J, Westra ER, Jackson RN, Wiedenheft B (2014) Unravelling the structural and mechanistic basis of CRISPR-Cas systems. Nat Rev Microbiol 12:479–492. https://doi.org/10.1038/nrmicro3279
Wick RR, Judd LM, Gorrie CL, Holt KE (2017) Unicycler: Resolving bacterial genome assemblies from short and long sequencing reads. PLoS Comput Biol 13:1–22. https://doi.org/10.1371/journal.pcbi.1005595
Acknowledgments
The authors thank the Núcleo de Plataformas Tecnológicas (NPT) from the Aggeu Magalhães Institute for the use of its sequencing platform.
Funding
This work was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico [Grant Number 404016/2016–2]; Fundação Oswaldo Cruz/ Conselho Nacional de Desenvolvimento Científico e Tecnológico [Grant Number: 400781/2019–0]; and Programa Inova Fiocruz – Geração de Conhecimento [Grant Number: VPPCB-007-FIO-18–2-99].
Author information
Authors and Affiliations
Contributions
Ana Carolina de Oliveira Luz: Conceptualization, methodology, investigation, data curation, writing (original draft), and writing (review and editing). Wilson José da Silva Junior: Methodology, software, and formal analysis. José Bandeira do Nascimento Junior: Methodology, software, and formal analysis. Julia Mariana Assis da Silva: Methodology and investigation. Valdir de Queiroz Balbino: Writing (review and editing) and supervision. Tereza Cristina Leal-Balbino: Conceptualization, writing (original draft), writing (review and editing), supervision, project administration, and funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interests.
Ethics approval
Not applicable. The present work did not involve humans, human specimens, or tissues. Bacteria were obtained purified from the hospitals per request.
Additional information
Communicated by Michael Polymenis.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
de Oliveira Luz, A.C., da Silva Junior, W.J., do Nascimento Junior, J.B. et al. Genetic characteristics and phylogenetic analysis of Brazilian clinical strains of Pseudomonas aeruginosa harboring CRISPR/Cas systems. Curr Genet 67, 663–672 (2021). https://doi.org/10.1007/s00294-021-01173-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00294-021-01173-4