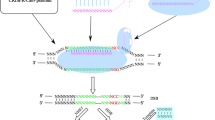
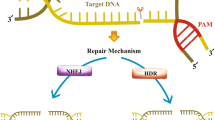
Abstract
The recent introduction of clustered regularly interspaced short palindromic repeats (CRISPR) and CRISPR associated protein (Cas) systems, offer an array of genome and transcriptome editing tools for clinical repair strategies. These include Cas9, Cas12a, dCas9 and more recently Cas13 effectors. RNA targeting CRISPR-Cas13 complexes show unique characteristics with the capability to engineer transcriptomes and modify gene expression, providing a potential clinical cancer therapy tool across various tissue types. Cas13 effectors such as RNA base editing for A to I replacement allows for precise transcript modification. Further applications of Cas13a highlights its capability of producing rapid diagnostic results in a mobile platform. This review will focus on the adaptions of existing CRISPR-Cas systems, along with new Cas effectors for transcriptome or RNA modifications used in disease modelling and gene therapy for haematological malignancy. We also address the current diagnostic and therapeutic potential of CRISPR-Cas systems for personalised haematological malignancy.


Similar content being viewed by others
References
Abudayyeh, O. O., Gootenberg, J. S., Konermann, S., Joung, J., Slaymaker, I. M., Cox, D. B., Zhang, F. (2016). C2c2 is a single-component programmable RNA-guided RNA-targeting CRISPR effector. Science, 353(6299), aaf5573. doi:https://doi.org/10.1126/science.aaf5573
Abudayyeh OO, Gootenberg JS, Essletzbichler P, Han S, Joung J, Belanto JJ, Zhang F (2017) RNA targeting with CRISPR-Cas13. Nature 550(7675):280–284. https://doi.org/10.1038/nature24049
Anzalone AV, Randolph PB, Davis JR, Sousa AA, Koblan LW, Levy JM, Liu DR (2019) Search-and-replace genome editing without double-strand breaks or donor DNA. Nature 576(7785):149–157. https://doi.org/10.1038/s41586-019-1711-4
Bhaya D, Davison M, Barrangou R (2011) CRISPR-Cas systems in bacteria and archaea: versatile small RNAs for adaptive defense and regulation. Annu Rev Genet 45:273–297. https://doi.org/10.1146/annurev-genet-110410-132430
Bradshaw G, Lualhati RR, Albury CL, Maksemous N, Roos-Araujo D, Smith RA, Griffiths LR (2018) Exome sequencing diagnoses X-linked Moesin-associated immunodeficiency in a primary immunodeficiency case. Front Immunol 9:420. https://doi.org/10.3389/fimmu.2018.00420
Charlesworth CT, Camarena J, Cromer MK, Vaidyanathan S, Bak RO, Carte JM, Porteus MH (2018) Priming human repopulating hematopoietic stem and progenitor cells for Cas9/sgRNA gene targeting. Mol Ther Nucleic Acids 12:89–104. https://doi.org/10.1016/j.omtn.2018.04.017
Charlesworth CT, Deshpande PS, Dever DP, Camarena J, Lemgart VT, Cromer MK, Porteus MH (2019) Identification of preexisting adaptive immunity to Cas9 proteins in humans. Nat Med 25(2):249–254. https://doi.org/10.1038/s41591-018-0326-x
Chen YH, Xu LP, Chen H, Liu DH, Zhang XH, Han W, Huang XJ (2011) The efficacy and safety of second allogeneic hematopoietic stem cell transplantation for post-transplant hematologic malignancies relapse. Zhonghua Nei Ke Za Zhi 50(6):489–491. https://doi.org/10.3760/cma.j.issn.0578-1426.2011.06.011
Cheng AW, Wang H, Yang H, Shi L, Katz Y, Theunissen TW, Jaenisch R (2013) Multiplexed activation of endogenous genes by CRISPR-on, an RNA-guided transcriptional activator system. Cell Res 23(10):1163–1171. https://doi.org/10.1038/cr.2013.122
Chew, W. L. (2018). Immunity to CRISPR Cas9 and Cas12a therapeutics. Wiley Interdiscip Rev Syst Biol Med, 10(1). doi:https://doi.org/10.1002/wsbm.1408
Cong L, Ran FA, Cox D, Lin S, Barretto R, Habib N, Zhang F (2013) Multiplex genome engineering using CRISPR/Cas systems. Science 339(6121):819–823. https://doi.org/10.1126/science.1231143
Cooper ML, Choi J, Staser K, Ritchey JK, Devenport JM, Eckardt K, DiPersio JF (2018) An “off-the-shelf” fratricide-resistant CAR-T for the treatment of T cell hematologic malignancies. Leukemia 32(9):1970–1983. https://doi.org/10.1038/s41375-018-0065-5
Cox DBT, Gootenberg JS, Abudayyeh OO, Franklin B, Kellner MJ, Joung J, Zhang F (2017) RNA editing with CRISPR-Cas13. Science 358(6366):1019–1027. https://doi.org/10.1126/science.aaq0180
Doudna, J. A., and Charpentier, E. (2014). Genome editing. The new frontier of genome engineering with CRISPR-Cas9. Science, 346(6213), 1258096. doi:https://doi.org/10.1126/science.1258096
Doudna JA, Gersbach CA (2015) Genome editing: the end of the beginning. Genome Biol 16:292. https://doi.org/10.1186/s13059-015-0860-5
East-Seletsky A, O'Connell MR, Burstein D, Knott GJ, Doudna JA (2017) RNA targeting by functionally orthogonal type VI-A CRISPR-Cas enzymes. Mol Cell 66(3):373–383 e373. https://doi.org/10.1016/j.molcel.2017.04.008
Eid A, Alshareef S, Mahfouz MM (2018) CRISPR base editors: genome editing without double-stranded breaks. Biochem J 475(11):1955–1964. https://doi.org/10.1042/BCJ20170793
Elbashir SM, Lendeckel W, Tuschl T (2001) RNA interference is mediated by 21- and 22-nucleotide RNAs. Genes Dev 15(2):188–200. https://doi.org/10.1101/gad.862301
Fire AZ (2007) Gene silencing by double-stranded RNA. Cell Death Differ 14(12):1998–2012. https://doi.org/10.1038/sj.cdd.4402253
Fire A, Xu S, Montgomery MK, Kostas SA, Driver SE, Mello CC (1998) Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 391(6669):806–811. https://doi.org/10.1038/35888
Gasiunas G, Siksnys V (2013) RNA-dependent DNA endonuclease Cas9 of the CRISPR system: holy grail of genome editing? Trends Microbiol 21(11):562–567. https://doi.org/10.1016/j.tim.2013.09.001
Gasiunas G, Barrangou R, Horvath P, Siksnys V (2012) Cas9-crRNA ribonucleoprotein complex mediates specific DNA cleavage for adaptive immunity in bacteria. Proc Natl Acad Sci U S A 109(39):E2579-2586. https://doi.org/10.1073/pnas.1208507109
Gonzalez-Romero E, Martinez-Valiente C, Garcia-Ruiz C, Vazquez-Manrique RP, Cervera J, Sanjuan-Pla A (2019) CRISPR to fix bad blood: a new tool in basic and clinical hematology. Haematologica 104(5):881–893. https://doi.org/10.3324/haematol.2018.211359
Gootenberg JS, Abudayyeh OO, Kellner MJ, Joung J, Collins JJ, Zhang F (2018) Multiplexed and portable nucleic acid detection platform with Cas13, Cas12a, and Csm6. Science 360(6387):439–444. https://doi.org/10.1126/science.aaq0179
Henrickson, S. E., Andre-Schmutz, I., Lagresle-Peyrou, C., Deardorff, M. A., Jyonouchi, H., Neven, B., Heimall, J. R. (2019). Hematopoietic Stem Cell Transplant for the Treatment of X-MAID. Frontiers in Pediatrics, 7(170). doi:https://doi.org/10.3389/fped.2019.00170
Howlader N, Mariotto AB, Besson C, Suneja G, Robien K, Younes N, Engels EA (2017) Cancer-specific mortality, cure fraction, and noncancer causes of death among diffuse large B-cell lymphoma patients in the immunochemotherapy era. Cancer 123(17):3326–3334. https://doi.org/10.1002/cncr.30739
Hussaini MO, Mirza AS, Komrokji R, Lancet J, Padron E, Song J (2018) Genetic landscape of acute myeloid leukemia interrogated by next-generation sequencing: a large cancer center experience. Cancer Genomics Proteomics 15(2):121–126. https://doi.org/10.21873/cgp.20070
Jinek M, Chylinski K, Fonfara I, Hauer M, Doudna JA, Charpentier E (2012) A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 337(6096):816–821. https://doi.org/10.1126/science.1225829
Kaczmarek JC, Kowalski PS, Anderson DG (2017) Advances in the delivery of RNA therapeutics: from concept to clinical reality. Genome Med 9(1):60. https://doi.org/10.1186/s13073-017-0450-0
Kohn DB, Porteus MH, Scharenberg AM (2016) Ethical and regulatory aspects of genome editing. Blood 127(21):2553–2560. https://doi.org/10.1182/blood-2016-01-678136
Komor AC, Kim YB, Packer MS, Zuris JA, Liu DR (2016) Programmable editing of a target base in genomic DNA without double-stranded DNA cleavage. Nature 533(7603):420–424. https://doi.org/10.1038/nature17946
Konermann, S., Lotfy, P., Brideau, N. J., Oki, J., Shokhirev, M. N., and Hsu, P. D. (2018). Transcriptome Engineering with RNA-Targeting Type VI-D CRISPR Effectors. Cell, 173(3), 665–676 e614. doi:https://doi.org/10.1016/j.cell.2018.02.033
Lagresle-Peyrou, C., Luce, S., Ouchani, F., Soheili, T. S., Sadek, H., Chouteau, M., Andre-Schmutz, I. (2016). X-linked primary immunodeficiency associated with hemizygous mutations in the moesin (MSN) gene. J Allergy Clin Immunol, 138(6), 1681–1689 e1688. doi:https://doi.org/10.1016/j.jaci.2016.04.032
Legut M, Dolton G, Mian AA, Ottmann OG, Sewell AK (2018) CRISPR-mediated TCR replacement generates superior anticancer transgenic T cells. Blood 131(3):311–322. https://doi.org/10.1182/blood-2017-05-787598
Lin S, Staahl BT, Alla RK, Doudna JA (2014) Enhanced homology-directed human genome engineering by controlled timing of CRISPR/Cas9 delivery. Elife 3:e04766. https://doi.org/10.7554/eLife.04766
Liu X, Zhao Y (2018) CRISPR/Cas9 genome editing: fueling the revolution in cancer immunotherapy. Curr Res Transl Med 66(2):39–42. https://doi.org/10.1016/j.retram.2018.04.003
Liu, L., Li, X., Ma, J., Li, Z., You, L., Wang, J., Wang, Y. (2017). The Molecular Architecture for RNA-Guided RNA Cleavage by Cas13a. Cell, 170(4), 714–726 e710. doi:https://doi.org/10.1016/j.cell.2017.06.050
Montiel-Gonzalez MF, Vallecillo-Viejo IC, Rosenthal JJ (2016) An efficient system for selectively altering genetic information within mRNAs. Nucleic Acids Res 44(21):e157. https://doi.org/10.1093/nar/gkw738
Moses C, Nugent F, Waryah CB, Garcia-Bloj B, Harvey AR, Blancafort P (2019) Activating PTEN tumor suppressor expression with the CRISPR/dCas9 system. Mol Ther Nucleic Acids 14:287–300. https://doi.org/10.1016/j.omtn.2018.12.003
Myhrvold C, Freije CA, Gootenberg JS, Abudayyeh OO, Metsky HC, Durbin AF, Sabeti PC (2018) Field-deployable viral diagnostics using CRISPR-Cas13. Science 360(6387):444–448. https://doi.org/10.1126/science.aas8836
National Institute of Health U.S National Library of Medicine (2020) Clinical Trials. Retrieved from https://clinicaltrials.gov/ct2
Nelles DA, Fang MY, Aigner S, Yeo GW (2015) Applications of Cas9 as an RNA-programmed RNA-binding protein. BioEssays 37(7):732–739. https://doi.org/10.1002/bies.201500001
Nelles DA, Fang MY, O’Connell MR, Xu JL, Markmiller SJ, Doudna JA, Yeo GW (2016) Programmable RNA tracking in live cells with CRISPR/Cas9. Cell 165(2):488–496. https://doi.org/10.1016/j.cell.2016.02.054
O’Connell MR, Oakes BL, Sternberg SH, East-Seletsky A, Kaplan M, Doudna JA (2014) Programmable RNA recognition and cleavage by CRISPR/Cas9. Nature 516(7530):263–266. https://doi.org/10.1038/nature13769
Papaemmanuil E, Gerstung M, Bullinger L, Gaidzik VI, Paschka P, Roberts ND, Campbell PJ (2016) Genomic classification and prognosis in acute myeloid leukemia. N Engl J Med 374(23):2209–2221. https://doi.org/10.1056/NEJMoa1516192
Pasqualucci L, Dalla-Favera R (2018) Genetics of diffuse large B-cell lymphoma. Blood 131(21):2307–2319. https://doi.org/10.1182/blood-2017-11-764332
Peng R, Lin G, Li J (2016) Potential pitfalls of CRISPR/Cas9-mediated genome editing. FEBS J 283(7):1218–1231. https://doi.org/10.1111/febs.13586
Pubmed, N. C. B. I. (2013). CRISPR Cas9 Genome Editing 2013. Retrieved from https://pubmed.ncbi.nlm.nih.gov/?term=crispr+cas9+genome+editingandfilter=years.2013-2013andtimeline=expanded
Ran FA, Hsu PD, Wright J, Agarwala V, Scott DA, Zhang F (2013) Genome engineering using the CRISPR-Cas9 system. Nat Protoc 8(11):2281–2308. https://doi.org/10.1038/nprot.2013.143
Ren J, Liu X, Fang C, Jiang S, June CH, Zhao Y (2017) Multiplex genome editing to generate universal CAR T cells resistant to PD1 inhibition. Clin Cancer Res 23(9):2255–2266. https://doi.org/10.1158/1078-0432.CCR-16-1300
Shen B, Zhang W, Zhang J, Zhou J, Wang J, Chen L, Skarnes WC (2014) Efficient genome modification by CRISPR-Cas9 nickase with minimal off-target effects. Nat Methods 11(4):399–402. https://doi.org/10.1038/nmeth.2857
Skarnes WC, Pellegrino E, McDonough JA (2019) Improving homology-directed repair efficiency in human stem cells. Methods 164–165:18–28. https://doi.org/10.1016/j.ymeth.2019.06.016
Stadtmauer, E. A., Fraietta, J. A., Davis, M. M., Cohen, A. D., Weber, K. L., Lancaster, E., June, C. H. (2020). CRISPR-engineered T cells in patients with refractory cancer. Science, 367(6481). doi:https://doi.org/10.1126/science.aba7365
Su S, Zou Z, Chen F, Ding N, Du J, Shao J, Liu B (2017) CRISPR-Cas9-mediated disruption of PD-1 on human T cells for adoptive cellular therapies of EBV positive gastric cancer. Oncoimmunology 6(1):e1249558. https://doi.org/10.1080/2162402X.2016.1249558
Xia AL, He QF, Wang JC, Zhu J, Sha YQ, Sun B, Lu XJ (2019) Applications and advances of CRISPR-Cas9 in cancer immunotherapy. J Med Genet 56(1):4–9. https://doi.org/10.1136/jmedgenet-2018-105422
Yoshida M, Yokota E, Sakuma T, Yamatsuji T, Takigawa N, Ushijima T, Naomoto Y (2018) Development of an integrated CRISPRi targeting DeltaNp63 for treatment of squamous cell carcinoma. Oncotarget 9(49):29220–29232. https://doi.org/10.18632/oncotarget.25678
Zalatan JG, Lee ME, Almeida R, Gilbert LA, Whitehead EH, La Russa M, Lim WA (2015) Engineering complex synthetic transcriptional programs with CRISPR RNA scaffolds. Cell 160(1–2):339–350. https://doi.org/10.1016/j.cell.2014.11.052
Zhang Q, Rho M, Tang H, Doak TG, Ye Y (2013) CRISPR-Cas systems target a diverse collection of invasive mobile genetic elements in human microbiomes. Genome Biol 14(4):R40. https://doi.org/10.1186/gb-2013-14-4-r40
Zhang F, Wen Y, Guo X (2014) CRISPR/Cas9 for genome editing: progress, implications and challenges. Hum Mol Genet 23(R1):R40-46. https://doi.org/10.1093/hmg/ddu125
Zhang Y, Rajan R, Seifert HS, Mondragon A, Sontheimer EJ (2015) DNase H Activity of Neisseria meningitidis Cas9. Mol Cell 60(2):242–255. https://doi.org/10.1016/j.molcel.2015.09.020
Zimmerly S, Guo H, Eskes R, Yang J, Perlman PS, Lambowitz AM (1995) A group II intron RNA is a catalytic component of a DNA endonuclease involved in intron mobility. Cell 83(4):529–538. https://doi.org/10.1016/0092-8674(95)90092-6
Acknowledgments
This work was supported by Queensland University of Technology (QUT), Centre for Genomics and Personalised Health and the Genomics Research Centre. We also thank Gabrielle Bradshaw for her founding investigations into MSN mutations and Primary Immunodeficiency disorder and Nicholas Harvey for his insightful comments on the manuscript. This work was supported by philanthropic funding from the Herbert family and also by the Genomics Research Centre, Institute of Health and Biomedical Innovation, QUT. The authors declare they have no conflicts of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest and that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Human and animal rights
The research presented does not involve human participants and/or animals, and as such no informed consent was sought or received.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Elliott, E.K., Haupt, L.M. & Griffiths, L.R. Mini review: genome and transcriptome editing using CRISPR-cas systems for haematological malignancy gene therapy. Transgenic Res 30, 129–141 (2021). https://doi.org/10.1007/s11248-020-00232-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-020-00232-9